Why is bromination more selective?

Daniel Johnson
Published Mar 04, 2026
Why is bromination more selective?
Bromination of alkanes occurs by a similar mechanism, but is slower and more selective because a bromine atom is a less reactive hydrogen abstraction agent than a chlorine atom, as reflected by the higher bond energy of H-Cl than H-Br.
What is the main reason of using NBS in allylic bromination?
NBS As A Reagent For Allylic Bromination NBS is used as a substitute for Br2 in these cases since Br2 tends to react with double bonds to form dibromides. The advantage of NBS is that it provides a low-level concentration of Br2, and bromination of the double bond doesn’t compete as much.
Is allylic a markovnikov bromination?
In allylic bromination, the Br atom appears on the carbon next to the double bond: This reaction goes through a radical mechanism and it is interesting to notice the difference with the aniti-Markovnikov radical bromination: First, it is important to mention that allylic radicals are “very stable”.
What makes radical bromination a selective reaction?
The Transition State For Bromination Resembles The Products (A “Late” Transition State) Which Are Farther Apart In Energy. So Selectivity Is High. Here, it’s a “late” transition state, so the difference in activation energies between primary and secondary will closely resemble the differences in energy between the two.
What is reactivity selectivity principle explain?
In chemistry the reactivity–selectivity principle or RSP states that a more reactive chemical compound or reactive intermediate is less selective in chemical reactions. In this context selectivity represents the ratio of reaction rates.
What is the role of NBS?
N-Bromosuccinimide or NBS is a chemical reagent used in radical substitution, electrophilic addition, and electrophilic substitution reactions in organic chemistry. NBS can be a convenient source of Br•, the bromine radical.
What does NBS and peroxide do?
Alkenes react with NBS in dry CCl4 under reflux conditions to give allyl bromide. The reaction is initiated by light or peroxide. Although a number of reagents are available for bromination of allylic C-H bond of alkenes, NBS is most commonly used. The reaction is called Wohl-Zigler bromination.
What do you mean by allylic rearrangement?
Definition. An allylic rearrangement or allylic shift is an organic reaction in which the double bond in an allyl chemical compound shifts to the next carbon atom. It is encountered in nucleophilic substitution.
Which of the following reagent can be used for allylic bromination?
The brominating reagent, N-bromosuccinimide (NBS), has proven useful for achieving allylic or benzylic substitution in CCl4 solution at temperatures below its boiling point (77 ºC). One such application is shown in the second equation.
Is radical bromination selective?
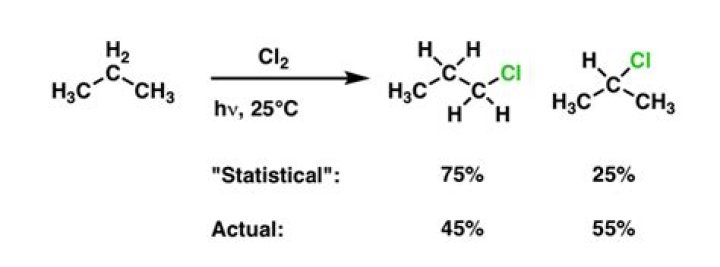
Chlorination. In last blog post on radicals we saw this data that compares the chlorination of propane vs. For bromination, the reaction is selective for secondary C-H over primary C-H by a factor of 97/(3/3) = 97 to 1. …
What is the difference between allylic and benzylic bromination?
Benzylic Bromination Allylic and benzylic bromination refer to the substituion of a bromine for a hydrogen on an allylic carbon (a carbon atom 1 bond from an alkene) or on a benzylic carbon (a carbon atom 1 bond from a benzene). These two reactions are carried out using N-bromosuccinimide (abbreviated NBS).
What is the driving force of allylic bromination?
This is the driving force of the allylic bromination. Step 1: The first step of allylic bromination is the homolytic cleavage of the N-Br bond (initiation) of the N -bromosuccinimide (NBS): Notice that the imide group can stabilize the radical by two additional resonance structures which helps to initiate the homolysis of the N-Br bond:
What is the difference between allylic bromination and aniti-Markovnikov bromization?
In allylic bromination, the Br atom appears on the carbon next to the double bond: This reaction goes through a radical mechanism and it is interesting to notice the difference with the aniti -Markovnikov radical bromination:
Why Br2 is not used for allylic bromination?
While Br2 is the standard reagent for free radical bromination, it is not the best choice for allylic bromination of an alkene or benzylic bromination of an aromatic compound due to the production of unwanted side products which decrease the yield of the desired product.



