Why do we use 3 electrode system?

Mia Smith
Published Feb 27, 2026
Why do we use 3 electrode system?
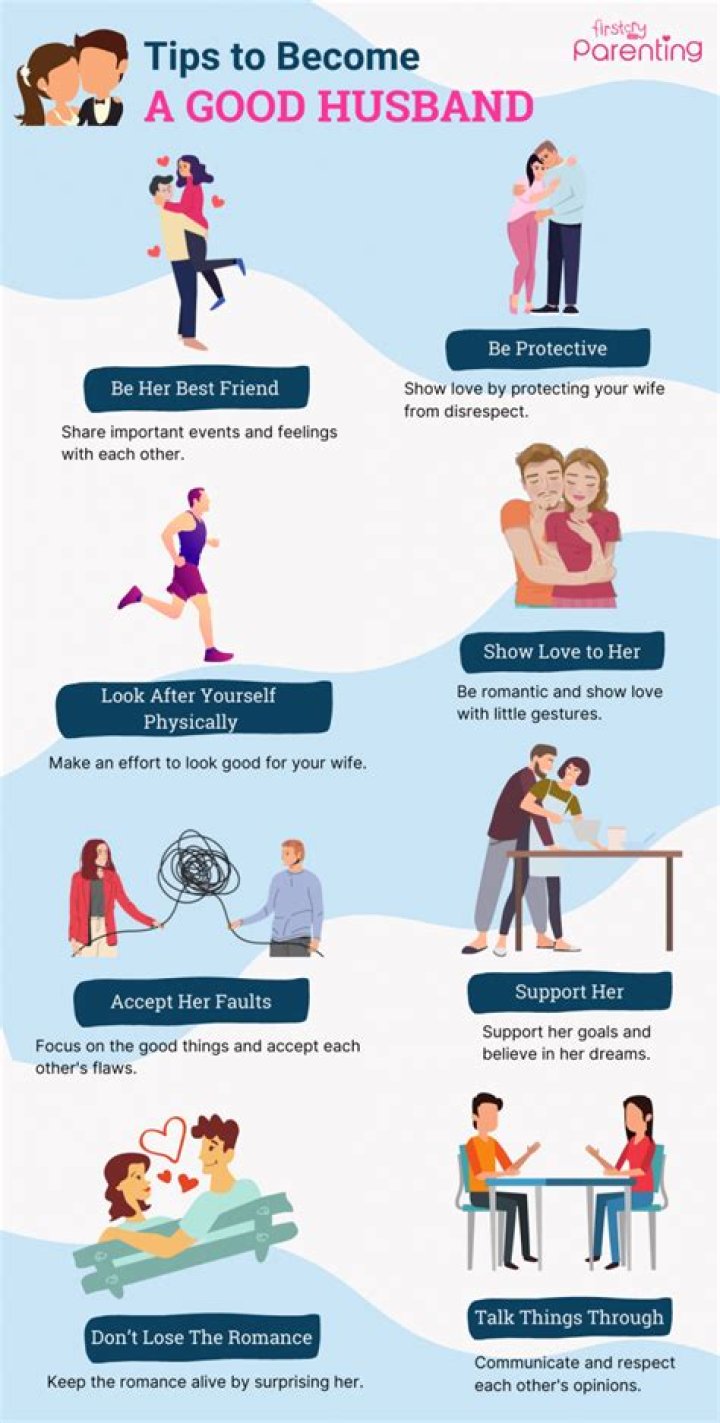
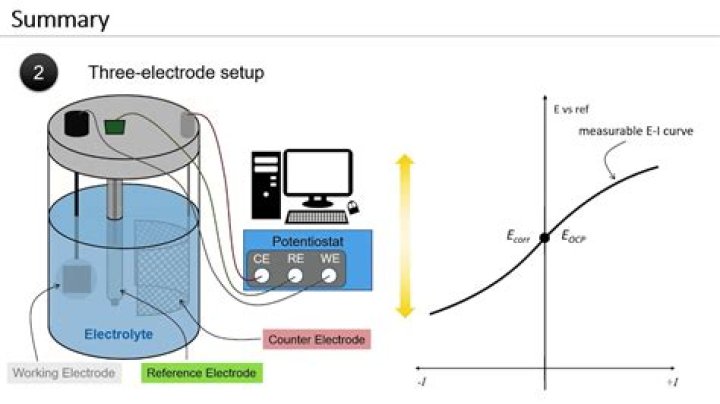
For a better control and measurement of the current and potential going through the cell during the electricity driven chemical reaction, it is better to use a three electrode system to reduce and compensate the potential changes caused by large currents passing through the working and counter electrodes.
Why do we use 3 electrodes in voltammetry?
You need a 3 electrode system, because the reference electrode must not take part in the redox reaction. Otherwise, the potential you’re measuring will be inaccurate. Therefore, the reference electrode should be a little further away from the WE or CE.
What is the difference between 2 electrode and 3 electrode system?
Three-electrode setups have a distinct experimental advantage over two-electrode setups: they measure only one half of the cell. That is, the potential changes of the working electrode are measured independent of changes that may occur at the counter electrode.
What is electrode system?
The working electrode is the electrode in an electrochemical system on which the reaction of interest is occurring. Depending on whether the reaction on the electrode is a reduction or an oxidation, the working electrode is called cathodic or anodic, respectively.
Why 3 electrode system is better than two electrode system?
3-electrode cell just gives you the electrochemical signature of the material by itself. A 2-electrode cell just gives you the current flowing between the 2 électrodes and none of the electrode potential is fixed so that you cannot know at whci potential (vs a reference) a reaction occurs.
What are the roles of the three electrodes working reference and auxiliary?
All Answers (16) Reference electrode allows you to measure the potential of the working electrode with out passing current through it while counter (auxiliary) electrode allows you to pass current.
How does a 3 electrode system work?
The three electrode system consists of a working electrode, counter electrode, and reference electrode. The reference electrode’s role is to act as a reference in measuring and controlling the working electrode potential, without passing any current.
What are the three types of electrodes?
Types of Electrodes: Gas electrode, metal metal salt electrode, etc.
Why three electrode system is better than two electrode system?
What is electrode in chemistry?
An electrode by definition is a point where current enters and leaves the electrolyte. When the current leaves the electrodes it is known as the cathode and when the current enters it is known as the anode. Electrodes are vital components of electrochemical cells.
Why do we need two electrodes?
This is because electrodes are substantially more conductive than the surrounding tissue. To record a biopotential signal, two electrodes are required. The pair of electrodes will measure the voltage which is impressed between the two equipotential areas created by the electrodes.
What is reference electrode in electrochemistry?
A reference electrode is an electrode which has a stable and well-known electrode potential. The simplest is when the reference electrode is used as a half-cell to build an electrochemical cell. This allows the potential of the other half cell to be determined.
Where are the electrodes placed for an EEG?
Electroencephalogram (EEG) electrodes are placed around the head region in order to gather electrical impulses from the brain and direct these to a type of machine that is used as a diagnostic tool for analyzing brain activity. This brain activity measurement is recorded by a technique known as encephalography.
What is the standard electrode potential?
Standard Electrode Potentials . In an electrochemical cell, an electric potential is created between two dissimilar metals. This potential is a measure of the energy per unit charge which is available from the oxidation/reduction reactions to drive the reaction.
What is standard electrode?
Standard electrode potential. In electrochemistry , it is defined as the measure of the individual potential of reversible electrode at standard state with ions at an effective concentration of 1mol dm-3 at the pressure of 1 atm The basis for an electrochemical cell, such as the galvanic cell, is always a redox reaction which can be broken…
How does a Potentiostatic circuit work?
The potentiostat contains many internal circuits that allow it to function in this capacity. The circuits generate and measure potentials and currents. External wires in a cell cable connect the potentiostat circuit to the electrodes in the electrochemical cell.