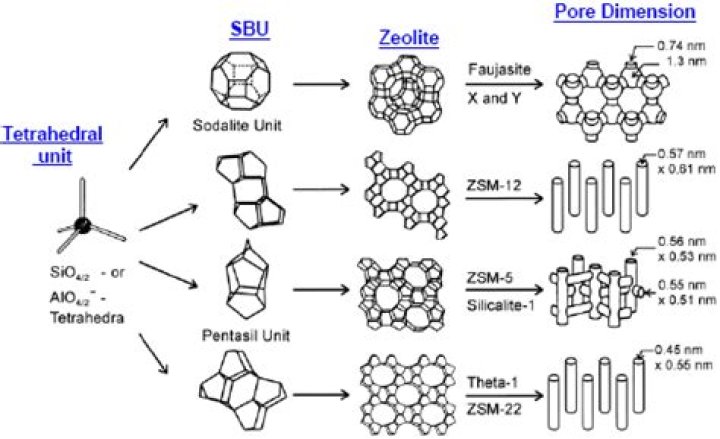
Why are zeolites good shape selective catalysts?

Michael Henderson
Published Mar 08, 2026
Why are zeolites good shape selective catalysts?
Answer: Zeolites have the ability to selectively adsorb molecules based on their size. This is based on the size of the porous structure of zeolites. Hence they are good shape selective catalysts.
Are zeolites shape selective catalysis?
At the end of the 1960s, the concept of shape-selective catalysis with zeolites was introduced to petrochemistry, and the zeolites became of increasing importance in catalysis research and applied catalysis. The shape selectivity of zeolites is based on the interaction of reactants with the well-defined pore system.
What is shape selectivity catalyst?
A catalytic reaction which depends upon the pore structure of the catalyst and on the size of the reactant and the product molecules is called shape-selective catalysis. For example, catalysis by zeolites is a shape-selective catalysis.
What is the important feature of zeolite catalysis?
For zeolites, the ability to exchange cations is another essential feature for applications in catalysis. As a result of their high surface areas, nanoporous catalysts contain a higher density of active sites that are directly involved in the reaction at a molecular level.
Which shape selective catalyst is used in getting petrol from alcohol?
Zeolite Socony Mobil-5, shortly known as ZSM-5 is a selective catalyst used to convert alcohol directly into gasoline.
Which statement is incorrect about zeolites?
Option B: In zeolites, some of the \[S{i^{4 + }}\] ions can even be replaced by \[A{l^{3 + }}\]. This results in the unbalanced anionic charge. And in order to maintain electrical neutrality, it becomes necessary to introduce the positive ions. Hence, the statement in Option B is incorrect.
What is shape selective catalyst example?
Answer: A catalytic reaction which depends upon the pore structure of the catalyst and on the size of the reactant and the product molecules is called shape-selective catalysis. For example, catalysis by zeolites is a shape-selective catalysis. The pore size present in the zeolites ranges from 260-740 pm.
Why is the shape of a catalyst important?
The pressure drop across a tube packed with catalyst depends on the shape of the catalyst. A smaller pressure drop is better. The smaller the ring the higher the catalytic activity and the better the heat transfer properties but the higher the pressure drop as the gases pass through the reformer tube.
What is shape selective catalysis examples?
A catalytic reaction that depends on the molecules of the product and on the size of the reactant and the catalyst’s pore structure is called shape-selective catalysis. Example: Example of shape-selective catalysis is the catalysis reaction by using zeolite.
How zeolite is effective as catalyst and how it can be controlled?
Zeolites can be shape-selective catalysts either by transition state selectivity or by exclusion of competing reactants on the basis of molecular diameter. They have also been used as oxidation catalysts. The reactions can take place within the pores of the zeolite, which allows a greater degree of product control.
Which type of catalyst converts alcohol directly into gasoline?
zeolite catalyst
Ethanol, methanol and larger alcohols can be converted into gasoline at 300-400°C in the presence of a zeolite catalyst (HZSM-5).
Do zeolites exist naturally?
Zeolites are microporous, aluminosilicate minerals commonly used as commercial adsorbents and catalysts. Zeolites occur naturally but are also produced industrially on a large scale. As of December 2018, 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known.