Why are the halogens most likely to react with the noble gases?

Michael Henderson
Published Mar 19, 2026
Why are the halogens most likely to react with the noble gases?
The number of electrons in the outermost shell of an atom determines its reactivity. Noble gases have low reactivity because they have full electron shells. Halogens are highly reactive because they readily gain an electron to fill their outermost shell.
How does the electron distribution differ in halogens?
Hydrogen has one electron in its electron shell, needing one additional electron to fill that shell. The halogens all have seven electrons in their outer electron shells. These electron shells all need eight electrons for completion, so the halogens are also missing a single electron.
What are the effects of halogens?
Exposure to halogens, such as chlorine or bromine, results in environmental and occupational hazard to the lung and other organs. Chlorine is highly toxic by inhalation, leading to dyspnea, hypoxemia, airway obstruction, pneumonitis, pulmonary edema, and acute respiratory distress syndrome (ARDS).
What is the trend of electron affinity in halogens?
Since the atomic size increases down the group, electron affinity generally decreases (At < I < Br < F < Cl). An electron will not be as attracted to the nucleus, resulting in a low electron affinity. However, fluorine has a lower electron affinity than chlorine.
How do the electron arrangements differ between the halogens and the noble gases?
Explanation: Halogens are very reactive because they have seven valence electrons and need one more to have eight valence electrons (an octet). The noble gases have filled valence shells as they occur in nature. Helium has a duet of valence electrons, and the rest of the noble gases have an octet.
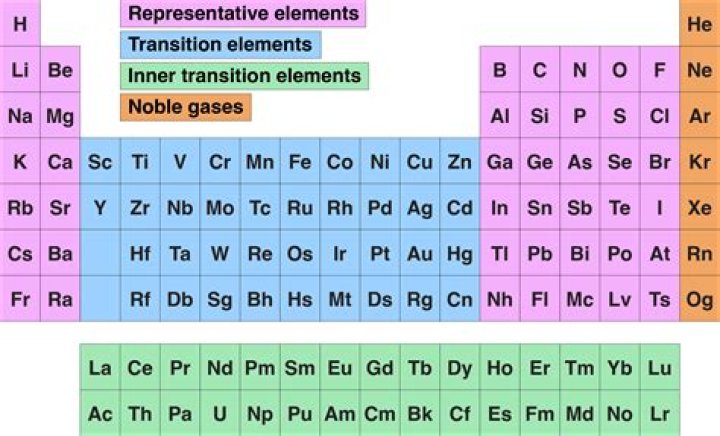
What is the difference between halogens and noble gas?
ii) Halogens exist as diatomic molecules when they are in their pure form while noble gases are monatomic. iii) Halogens are colored and have a distinctive and unpleasant odor while noble gases are colorless and odorless….Complete answer:
| Halogens | Noble gases |
|---|---|
| Very reactive | Non – reactive |
Why do halogens have the highest electron affinity?
The high electron affinities of the halogens are a result of their having an almost complete outer shell of electrons. Because of this overall attraction, energy will be released when the electron is captured by the fluorine atom. Similar reasoning also explains why oxygen also has a high electron affinity.
Why do halogens have similar chemical and physical properties?
Atoms of group 7 elements all have seven electrons in their outer shell. This means that the halogens all have similar chemical properties .
Why does chlorine increase Lipophilicity?
Hello, In general, to add halogen substituents (in particular Cl. Br, I) will increase the lipophilicity of your molecules: the element becomes bigger, is more polarized and the London dispersion forces are increased accordingly.
How do halogens affect the environment?
What’s more, halogens degrade air quality by promoting surface ozone formation. At ground-level, ozone is a pollutant (& greenhouse gas) and prolonged exposure can lead to respiratory ailments, including asthma, and is damaging to crops.
Why does electron affinity increase across a period and decrease down a group?
This is because as you go down the period table, new valence shells are added increasing the atomic radius. The new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the new electron decrease. Thus, electron affinity decreases.
What are the differences between halogens and noble gases?
Halogens are extremely reactive elements because they need one more electron to gain a full octet state while noble gases are extremely unstable because they already have their full octet state, hence they are placed in separate groups of the periodic table.