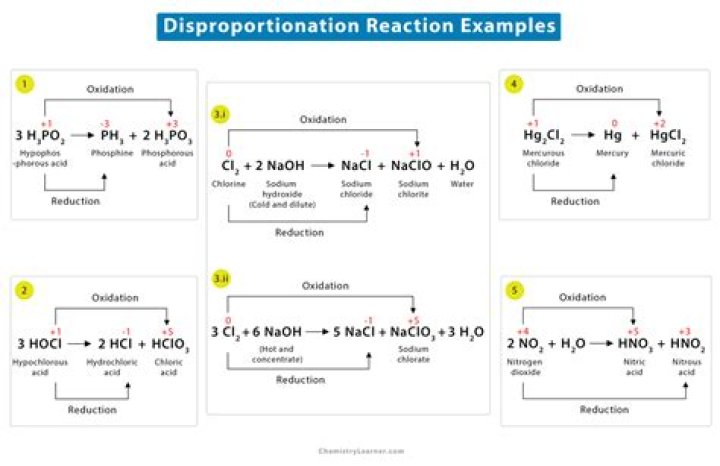
Which of the following is an example of disproportion reaction?

Sarah Cherry
Published Mar 12, 2026
Which of the following is an example of disproportion reaction?
2H2S+SO2→2H2O+3S.
Is F2 a disproportionation reaction?
It does not show positive oxidation state and does not undergo disproportionation reaction.
What do you mean by disproportionation reaction?
A Disproportionation reaction is a reaction in which a compound undergoes oxidation as well as reduction for example consider. 2H2O2=2H2O + O2. Here hydrogen peroxide is oxidized to oxygen and is reduced to water.
Does i2 disproportionate in water?
In a disproportionation reaction, the reactant takes part in redox reaxtion (both oxidation and reduction occurs simultaneously). Similarly, Br₂ and I₂ does not undergo disproportionation in water.
How do you know if a reaction is disproportionation?
Hint: A disproportionation reaction is when a substance is both oxidised and reduced forming different products. Check for the oxidation number of the elements in compounds and compare them on both sides.
Is 2f2 2oh a disproportionation reaction?
No because in the product, In both the compounds F- and OF2 , Fluorine’s oxidation state is -1. Hence Fluorine doesn’t disproportionate. Oxygen also, Initially has -2 oxidation state, but in the product its oxidation state becomes +2 in OF2 but still is -2 in H2O.
What is disproportionation reaction write between cl2 and oh?
Cl 2 is both an oxidizing and a reducing agent (disproportionation (dismutation)). ; Greenish-yellow gas with a pungent, irritating odor. [Note: Shipped as a liquefied compressed gas.] Search by reactants (Cl 2, OH -)
What is disproportionation give an example?
A disproportionation reaction is when a multiatomic species whose pertinent element has a specific oxidation state gets oxidized and reduced in two separate half-reactions, yielding two other products containing the same pertinent element. A convenient example is Mn2O3 becoming Mn2+ and MnO2 .
What is meant by disproportionation give two examples?
Disproportionation reactions are those in which the same substance undergoes oxidation as well as reduction, i.e., oxidation number of an element increases as well decreases to form two different products. e.g., (+3MnO2-4+4Hp+→+72MnOɵ4++4MnO2+2H2O)
Does P4 disproportionate?
The reaction, P4 + 3NaOH + 3H2O→ 3NaH2PO2 + PH3 , is a disproportionation reaction.
Does co show disproportionation reaction?
According to SEM, CO disproportionation reaction has resulted in carbon deposition in both amorphous and nanostructured forms. It was shown that preparative technique has significant effect on catalytic performance of iron-containing catalysts.
What is disproportionation reaction with Example Class 11?
The same element is oxidized and reduced in the disproportionation reaction. The example of disproportionation reaction is as follows: When H3PO3 is heated it undergoes a disproportionation reaction. During the disproportionation reaction, H3PO3 undergoes oxidation as well as reduction.
What are some examples of disproportionation reactions in organic chemistry?
Disproportionation reaction examples Dissociation of hydrogen peroxide. Dissociation of hydrogen peroxide is a disproportionation reaction. The oxygen atom in H 2 O 2 is in -1 oxidation state. It is oxidized to O 2 (ox.st = 0) as well as reduced to H 2 O (ox.st = -2).
What is disproporionation reaction?
In a disproportionation reaction, the same element is simultaneously oxidized and reduced. The reactions in which single reactant is oxidized and reduced is known as Disproporionation reactions is also a disproportionation reaction because O is reduced in forming H ₂ O and oxidized in forming O ₂
What is disproportionation in science?
She has taught science courses at the high school, college, and graduate levels. Disproportionation is a chemical reaction, typically a redox reaction, where a molecule is transformed into two or more dissimilar products. In a redox reaction, the species is simultaneously oxidized and reduced to form at least two different products.
What is the difference between disproportionation and comproportionation?
where A, A’, and A” are all different chemical species. The reverse reaction of disproportionation is called comproportionation. Examples: Hydrogen peroxide converting into water and oxygen is a disproportionation reaction.



