Which form of cyclodextrin is more soluble in water?

Daniel Johnson
Published Feb 10, 2026
Which form of cyclodextrin is more soluble in water?
The α-cyclodextrin ring is more strained, resulting in these hydroxyl groups being further apart and more able to interact with water, providing greater solubility.
How does cyclodextrin increase solubility?
Cyclodextrins and their derivatives, by forming guest- host complexes, encapsulate an insoluble drug and increase its aqueous solubility. The cyclodextrin carries the drug through the aqueous solution and the complex dissociates upon reach- ing the site of action.
How can cyclodextrins increase the solubility of a poorly water soluble drug?
Introduction. Cyclodextrin (CD) inclusion complexation, which is the formation of host–guest inclusion complexes by weak intermolecular interaction, has been shown to be a promising technique in enhancing solubility and bioavailability of poorly water soluble drugs [1].
Is cyclodextrin soluble in methanol?
HPCD readily dissolved not only in water but also in solvents such as methanol, ethanol, DMF, and DMSO.
What is 2 hydroxypropyl B Cyclodextrin?
2-Hydroxypropyl-β-cyclodextrin (HP-β-CD) is a chemically modified cyclic oligosaccharide produced from starch that is commonly used as an excipient. Although HP-β-CD has been suggested as a potential adjuvant for vaccines, its immunological properties and mechanism of action have yet to be characterized.
What is 2 hydroxypropyl Cyclodextrin?
Product Description. 2-Hydroxypropyl-β-cyclodextrin (HP-β-CD) is a cyclic oligosaccharide containing seven D-(+)-glucopyranose units that is widely used to improve the aqueous solubility of various compounds, especially those containing a phenyl group.
How does complexation increase solubility?
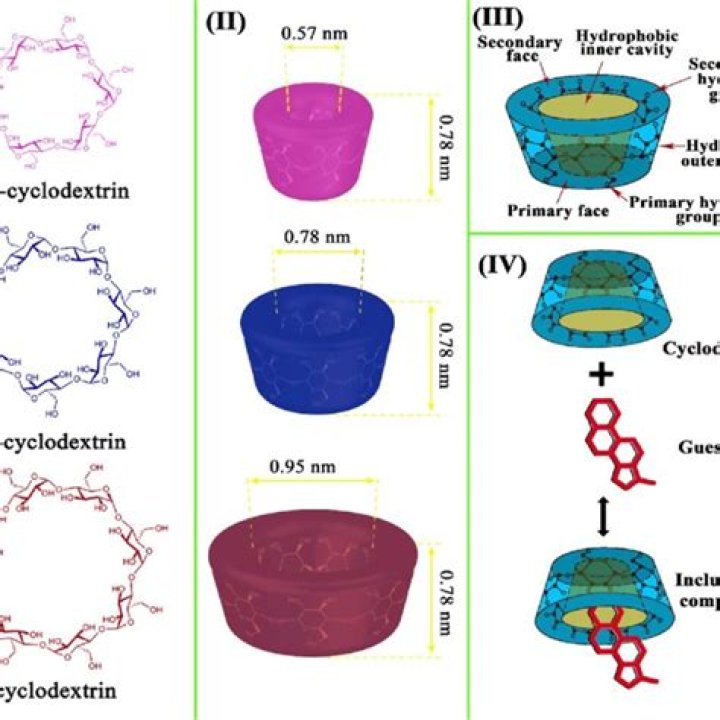
Their structural organization consists of a hydrophilic outer surface, as well as a hydrophobic inner cavity that allows inclusion of hydrophobic drug molecules. Cyclodextrin complexation leads to a significant increase in water solubility of the drug compared to uncomplexed drug (Sosnik et al., 2010).
Which of the following forms inclusion complex and improve aqueous solubility and dissolution rate?
Capsules with bigger particles and intense packing have poor drug release and dissolution rate due toan increase in the pore size….
| Q. | Which of the following forms inclusion complex and improves aqueous solubility and dissolution rate? |
|---|---|
| B. | PEG |
| C. | PVT |
| D. | Poloxamer |
| Answer» a. Cyclodextrins |
How we can enhance the solubility of drugs by complexation?
Complexation with CDs by different methods like physical mixing, melting, kneding, spray drying, freeze drying, co-evaporation has been reported to enhance the solubility, dissolution rate and bioavability of poorly water soluble drugs.
Which type of complex is formed by cyclodextrins?
inclusion complex
Cyclodextrin can form inclusion complex with a wide variety of solid, liquid and gaseous compounds.
Is cyclodextrin soluble in ethanol?
Moreover, it is slightly soluble in ethanol (0.1 g/100 mL).
Is cyclodextrin soluble in acetone?
as beta cyclodextrin and its other forms are highly hydrophilic in nature, it solubilize in different aqueous media. beta cyclodextrins & hydroxypropyl beta cyclodextrin are also soluble in number of polar aprotic solvent such as: tetrahydrofuran, dimethyl formamide, acetone, acetonitrile etc.
What is the solubility of cyclodextrin in water?
In the presence of urea, the solubility of β-cyclodextrin increases to 250 g/L. 38 At high pH, ionization of hydroxyl groups increases water solubility; at pH 12.5 the solubility of β-cyclodextrin is 750 g/L. 38,39 Cyclodextrins are insoluble in most organic solvents (Table 22.3).
What is the molecular weight of alpha-cyclodextrin?
alpha-CYCLODEXTRIN PubChem CID 444913 Synonyms alpha-CYCLODEXTRIN Alfadex Cyclohexapent Molecular Weight 972.8 Date s Modify 2021-08-28 Create 2005-06-24
What is the difference between alpha beta and gamma cyclodextrin in water?
The solubility of alpha, beta and gamma cyclodextrin in water is very different. Gamma cyclodextrin is the best water soluble, followed by alpha, beta cyclodextrin has the lowest solubility in water. The difference of the solubility of cyclodextrin is mainly related to its molecular structure.
How stable are cyclodextrins in the presence of bases?
Cyclodextrins are stable in the presence of bases. The differential scanning calorimetry (DSC) thermograms for α-, β- and γ-cyclodextrins are identical. Two heat absorption peaks are present: the first occurs at 100°C as water is evaporated from the crystals; the second, occurring at ∼250°C, is a result of crystal melting and thermal decomposition.