Which catalyst is used for hydrosilylation reaction?

Rachel Hickman
Published Mar 20, 2026
Which catalyst is used for hydrosilylation reaction?
For many years, platinum has been the metal of choice for designing hydrosilylation catalysts, Speier’s, Karstedt’s or Markó’s catalysts being the most repre- sentative examples (Figure 1). Other noble transition metals, such as rhodium, ruthenium, iridium or palladium, have also been used in this transformation [3].
How is platinum used as a catalyst?
Platinum works as a catalyst by collecting oxygen atoms (O), and letting them bind with the toxic carbon monoxide (CO), to create the less harmful carbon dioxide (CO2). This provides for the first time a good explanation for the high catalytic activity of platinum in oxidation reactions.
In what reaction does platinum become a catalyst?
Platinum is used in hydrogenation reactions. The most commonly used catalytic systems for chemical synthesis are PtO2, Pt/C, and Pt/Al2O3.
Why platinum is not used as catalyst?
Platinum is different because it tends not to become totally or irreversibly poisoned, i.e. sulfur- containing molecules inhibit rather than poison platinum-based catalysts (see also (2)).
What is Speier catalyst?
It is proposed that the very important and useful “Speier catalyst” contains the complex H[(C3H6)PtCl3] in a solution of isopropyl alcohol. It is this platinum complex which is the active catalyst ingredient in hydrosilylation reactions.
What is platinum rhodium catalyst?
Rhodium-platinum oxide (Rh–Pt oxide), or Nishimura’s catalyst, is an inorganic compound used as a hydrogenation catalyst. …
Is platinum the best catalyst?
Platinum and platinum alloys are the most efficient catalysts for speeding up chemical reactions in hydrogen fuel cells. Platinum is the only metal that can withstand the acidic conditions inside such a cell, but it is expensive, and this has limited the broad, large-scale applications of fuel cells.
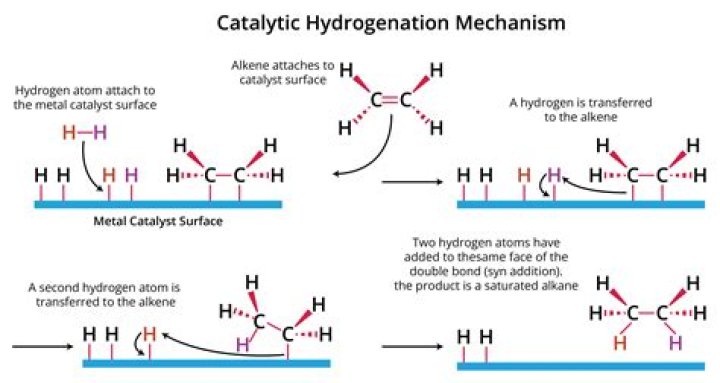
Which catalyst is used for hydrogenation of alkenes?
The Catalyst Catalysts commonly used in alkene hydrogenation are: platinum, palladium, and nickel. The metal catalyst acts as a surface on which the reaction takes place.
Why is platinum a good catalyst for fuel cells?
A good catalyst should be able to split up oxygen molecules into atoms but should not bind too strongly with the free atoms; the shorter distance between platinum atoms in the new material makes it a more effective catalyst because it binds even more weakly with the oxygen atoms.
Why is platinum the best catalyst?
Is platinum in a catalytic converter?
The catalyst component of a catalytic converter is usually platinum (Pt), along with palladium (Pd), and rhodium (Rh). All three of these platinum group metals, or PGMs, are extremely rare but have a broad range of applications in addition to catalytic converters.
What is the formula of Wilkinson’s catalyst?
C54H45ClP3Rh
Wilkinson’s catalyst/Formula



