What type of unit cell is ZnS?

Emma Martin
Published Mar 10, 2026
What type of unit cell is ZnS?
cubic unit cell
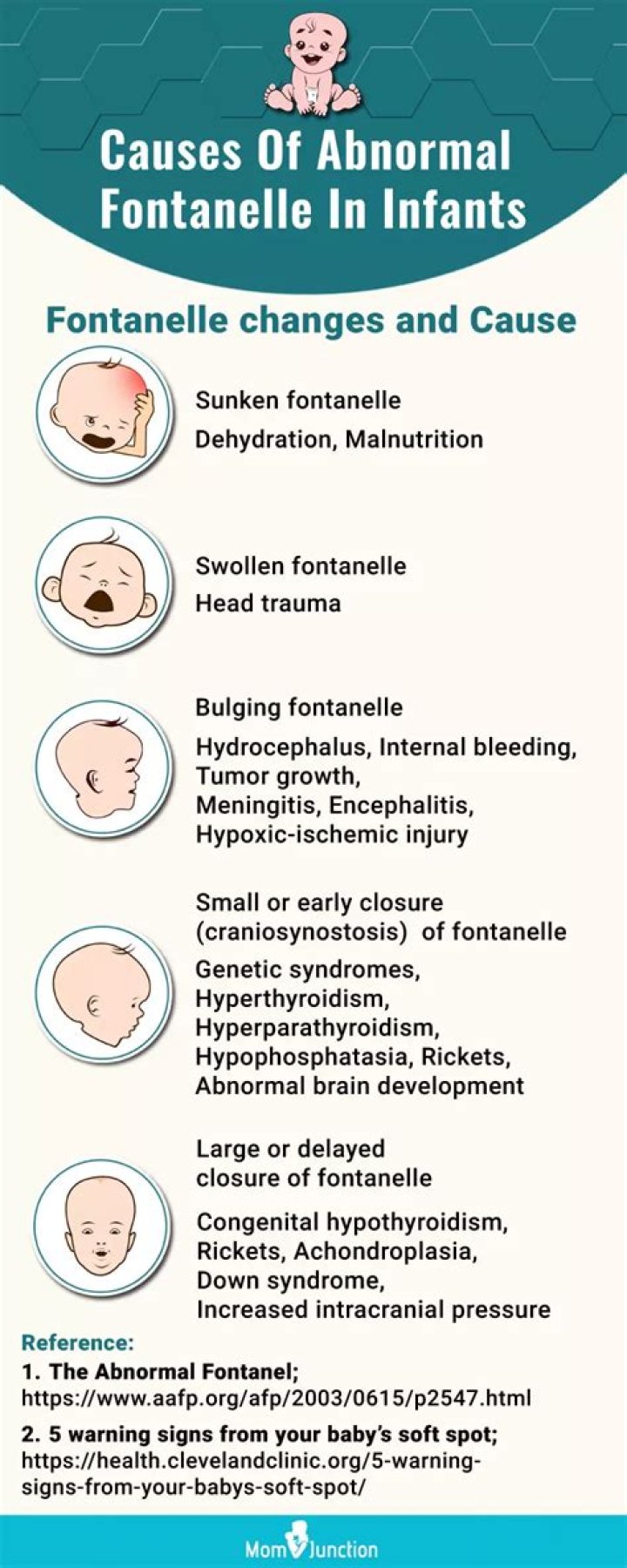
ZnS crystallizes in a cubic unit cell. The cell-edge length is 0.5411 nm.
Is zinc sulfide FCC or BCC?
Zincblende/sphalerite is based on a fcc lattice of anions whereas wurtzite is derived from an hcp array of anions. In both structures, the cations occupy one of the two types of tetrahedral holes present.
Is zinc sulfide A FCC?
ZnS, Zinc Sulphide, Zinc Blend, Wurtzite In the zinc blend structure the sulphur ions form an fcc structure and the zinc ions occupy half of the tetrahedral sites in this structure to attain charge neutrality. The crystal has a lattice parameter of 0.541 nm.
What type of crystal is ZnS?
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite….Zinc sulfide.
| Names | |
|---|---|
| Refractive index (nD) | 2.3677 |
| Structure | |
| Crystal structure | see text |
| Coordination geometry | Tetrahedral (Zn2+) Tetrahedral (S2−) |
Is ZnS ionic or covalent?
ZnS (zinc sulfide) is an ionic compound. This is because ionic compounds are made of metals and nonmetals.
Is ZnS Network Covalent?
ZnS is a / an ionic ionic crystal.
How many atoms are in a unit cell of ZnS?
Since the number of atoms in a single unit cell of Zn and S is the same, it is consistent with the formula ZnS….Introduction.
| Site | Zn | S |
|---|---|---|
| Total | 4 | 4 |
Is ZnS covalent or ionic?
Is ZnS aqueous or solid?
Zinc sulfide appears as a yellowish-white powder in a liquid. Insoluble in water and denser than water.
Is ZnS a molecular or ionic compound?
zinc sulfide
ZnS (zinc sulfide) is an ionic compound. This is because ionic compounds are made of metals and nonmetals.
Is ZnS an ionic crystal?
This is an ionic compound. This structure consists of two different types of atoms.
Is ZnS a molecular solid?
In the case of zinc sulphide, It is a metal sulphide having crystalline structure. It is not an example of molecular solid.
What is FCC unit cell in zinc sulfide?
They can be described by FCC unit cells with cations in the octahedral holes. The cubic form of zinc sulfide, zinc blende, also crystallizes in an FCC unit cell, as illustrated in the figure below. This structure contains sulfide ions on the lattice points of an FCC lattice.
What is the structure of zinc sulfide?
The cubic form of zinc sulfide, zinc blende, also crystallizes in an FCC unit cell, as illustrated in the figure below. This structure contains sulfide ions on the lattice points of an FCC lattice. (The arrangement of sulfide ions is identical to the arrangement of chloride ions in sodium chloride.)
How many zinc ions are there in ZnS?
The radius of a zinc ion is only about 40% of the radius of a sulfide ion, so these small Zn 2+ ions are located in alternating tetrahedral holes, that is, in one half of the tetrahedral holes. There are four zinc ions and four sulfide ions in the unit cell, giving the empirical formula ZnS.
What are some examples of polymorphism in zinc sulfide?
Wurtzite, the less common polymorph of zinc sulfide. ZnS exists in two main crystalline forms, and this dualism is often a salient example of polymorphism. In each form, the coordination geometry at Zn and S is tetrahedral.