What makes something more Bronsted acidic?

Rachel Hickman
Published Feb 17, 2026
What makes something more Bronsted acidic?
A Brønsted-Lowry acid is any species that is capable of donating a proton— H+start text, H, end text, start superscript, plus, end superscript. Strong acids and bases ionize completely in aqueous solution, while weak acids and bases ionize only partially.
How do you rank Bronsted acidity?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).
Is H2F a strong acid?
H2F is more acidic as the answer.
Why does proton acidity increase along a row in the periodic table?
In each vertical row of the periodic table, acidity usually increases with increasing atomic number because the valence (bonding) electrons are farther and farther away from the nucleus and less strongly attracted to the positive nucleus.
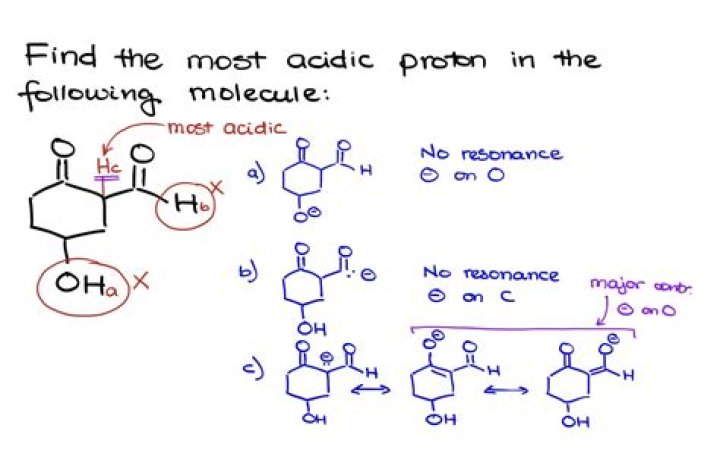
How does resonance increase acidity?
Resonance may delocalize this the electron pair that the base might use to form the new bond with the proton. This delocalization increases the stability of the base. Since a weaker base has a stronger conjugate acid, a compound whose conjugate base enjoys resonance stabilization will be more acidic.
What makes an acid acidic?
An acid is a substance that donates hydrogen ions. Because of this, when an acid is dissolved in water, the balance between hydrogen ions and hydroxide ions is shifted. Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic.
How do you find the strongest Bronsted-Lowry acid?
The strongest acids are at the bottom left, and the strongest bases are at the top right. The conjugate base of a strong acid is a very weak base, and, conversely, the conjugate acid of a strong base is a very weak acid.
What is the strongest Bronsted base?
When the acid is strong, the corresponding conjugate base will be weak as acids give up their protons, because the resulting conjugate base becomes more stable and does not react. So, the strongest bronsted base is $Cl{O^ – }$ .
Is H2CO3 strong or weak acid?
H2CO3 is a weak acid that dissociates into a proton (H+ cation) and a bicarbonate ion (HCO3- anion). This compound only partly dissociates in aqueous solutions. Furthermore, the conjugate base of carbonic acid, which is the bicarbonate ion, is a relatively good base.
Is H2SO4 a weak acid?
Weak acids are only slightly ionized. Phosphoric acid is stronger than acetic acid and so is ionized to a greater extent. Acetic acid is stronger than carbonic acid, and so on….Strong and Weak Acids and Acid Ionization Constant.
| Acid | Conjugate Base |
|---|---|
| H2SO4 (sulfuric acid) | HSO4− (hydrogen sulfate ion) |
| HNO3 (nitric acid) | NO3− (nitrate ion) |
| Weak Acids |
Why does acidity increase across a row?
Electronegativity Across a Period Across a period, more electronegative atoms are associated with stronger Brønsted acids. In other words, as we move to the right across the periodic table, acidity increases (all other things being equal).
Why does period increase acidity?
1. Going across a period the acid strength increases as there is an increase in electronegativity and the molecule gets more polar, with the hydrogen getting a larger partial positive charge. This makes it easier to heterlytically cleave the E-H bond to produce a stable anion.
What is the difference between Bronsted and Lewis acids and bases?
Acids and Bases: Lewis vs. Bronsted. There are two complementary definitions of acids and bases that are important: the Bronsted (or Bronsted-Lowry) definition: an acid is a proton (H+ ion) donor, and a base is a proton acceptor; the Lewis definition: an acid is an electron acceptor, and a base is an electron donor.
How are acids and bases defined in the Brønsted–Lowry theory?
In the Brønsted–Lowry theory acids and bases are defined by the way they react with each other, which allows for greater generality. The definition is expressed in terms of an equilibrium expression acid + base ⇌ conjugate base + conjugate acid.
What is the Lewis theory of acid base reactions?
In the same year that Brønsted and Lowry published their theory, G. N. Lewis proposed an alternative theory of acid–base reactions. The Lewis theory is based on electronic structure. A Lewis base is defined as a compound that can donate an electron pair to a Lewis acid, a compound that can accept an electron pair.
What is the formula for conjugate base of Brønsted acids?
Typical Brønsted Acids and Their Conjugate Bases Compound Ka pKa ConjugateBase Kb pKb HI 3 x 109 -9.5 I- 3 x 10-24 23.5 HCl 1 x 106 -6 Cl- 1 x 10-20 20