What macromolecules have hydroxyl groups

Sarah Cherry
Published Apr 15, 2026
Hydroxyl groups appear on carbohydrates (A glucose), on some amino acids (B Threonine), and on nucleic acids (C Adenine).
What are hydroxyl groups found in?
Hydroxyl groups are very common in biological molecules. Hydroxyl groups appear on carbohydrates (A), on some amino acids (B), and on nucleic acids (C).
Do proteins have hydroxyl groups?
Amino acids are the function units of protein. Each amino acid molecule contains a carboxyl group at one end, and an amino group at the other end. … When the protein is formed, the carboxyl group loses the hydroxyl group attached to it, while the amino group loses a hydrogen.
Are hydroxyl groups found in lipids?
Although the compositions of lipids vary, the most common functional groups are ester (both carboxylate and phosphate) and alcohol groups. … Carboxylate and phosphate groups are found in phospholipids such as lecithin. Amido, phosphate, and hydroxyl groups are found in sphingolipids such as sphingomyelin.What functional groups are in macromolecules?
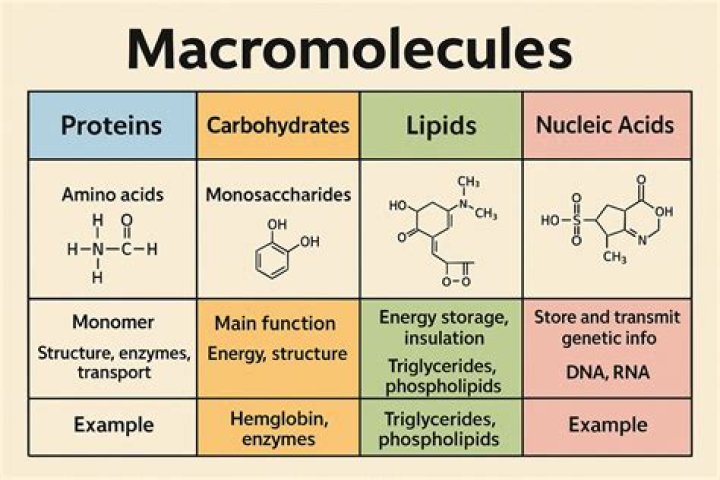
Functional groups in biological molecules play an important role in the formation of molecules like DNA, proteins, carbohydrates, and lipids. Functional groups include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl.
How many hydroxyl groups are present in?
Five OH groups are present: (Structure is only for reference.)
What compound has a hydroxyl group?
Organic compounds with a hydroxyl group are called alcohols. Their names often end with –ol. In enols, the carbon atom with the hydroxyl group is connected to the neighboring carbon atom with a double bond. Phenols are benzene rings with an -OH group.
Where are phospholipids found?
Where are phospholipids found? Each one of your cells is encased in a lipid layer called the cell membrane. The health of our cell membrane and the specific phospholipid content of it is integral to cellular metabolism.What macromolecules are sulfhydryl found on?
The correct answer is (a) proteins. Many different amino acids, which make up proteins, have sulfhydryl groups in their variable portions.
Are triglycerides phospholipids?Phospholipids are similar to triglycerides, but they do vary slightly in form and function. While triglycerides have glycerol and three fatty acids, phospholipids have glycerol, two fatty acids and a phosphate. Phosphates are molecules with charges and have oxygen and phosphorus. … Fat cells do not store phospholipids.
Article first time published onWhich amino acids contain a hydroxyl group?
Two amino acids, serine and threonine, contain aliphatic hydroxyl groups (that is, an oxygen atom bonded to a hydrogen atom, represented as ―OH). Tyrosine possesses a hydroxyl group in the aromatic ring, making it a phenol derivative.
Is COOH a hydroxyl group?
carboxylic acid, any of a class of organic compounds in which a carbon (C) atom is bonded to an oxygen (O) atom by a double bond and to a hydroxyl group (―OH) by a single bond. … The carboxyl (COOH) group is so-named because of the carbonyl group (C=O) and hydroxyl group.
What is a hydroxyl group quizlet?
hydroxyl group: structure. —OH : A hyrdrogen atom bonded to an oxygen atom, bonded to the carbon skeleton of the organic molecule. hydroxyl group: compound name. Alcohols – names usually end in “-ol”
Are hydroxyl groups hydrophobic?
Alcohols. Hydroxyl groups (-OH), found in alcohols, are polar and therefore hydrophilic (water liking) but their carbon chain portion is non-polar which make them hydrophobic.
How do you identify a hydroxyl group?
A hydroxyl group consists of one hydrogen and one oxygen atom and can be written as either -OH or HO-. Hydroxyl groups are polar, and the oxygen side is always negative, while the hydrogen side is always positive.
Is the hydroxyl group polar or nonpolar?
Functional GroupPropertiesHydroxylPolarMethylNonpolarCarbonylPolar
What makes a hydroxyl group?
Hydroxyl groups are simple structures consisting of an oxygen atom with two lone pairs bonded to a hydrogen atom. … The addition of a hydroxyl group converts many organic compounds into alcohols, enhancing their solubility in water.
What is a hydroxyl group in DNA?
While DNA contains deoxyribose, RNA contains ribose, characterised by the presence of the 2′-hydroxyl group on the pentose ring (Figure 5). This hydroxyl group make RNA less stable than DNA because it is more susceptible to hydrolysis.
Is hydroxyl group ionic?
Hydroxide ion and hydroxy group are nucleophiles and can act as catalysts in organic chemistry. Many inorganic substances which bear the word hydroxide in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxy groups.
How many hydroxyl groups are present in glucose?
From the figure, it is understood that glucose has five hydroxyl groups which are attached to the carbon atom. An aldehyde group is attached to the sixth carbon atom.
Which of the following does not contains hydroxyl group?
Ammonia is a base but it does not contain hydroxyl group because on reacting with water ammonia forms ammonium hydroxide which further on ionization gives ammonium ion and hydroxide ion. The reaction of ammonia is shown below. The ionization reaction of ammonium hydroxide is shown below.
Is a hydroxyl group the same as an alcohol group?
Hydroxyl group is a functional group present in organic and inorganic compounds such as alcohols. … The main difference between hydroxyl and alcohol is that a hydroxyl is a functional group whereas alcohol is an organic compound.
Where is sulfhydryl found?
Sulfhydryl groups can be found in the amino acid cysteine. When two cysteine residues are in close proximity to each other, they can form a disulfide bridge also called cystine. The formation of a disulfide bond is an example of a post translational modification.
Are carbonyl groups in lipids?
Properties of Functional Groups Some of the important functional groups in biological molecules include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. These groups play an important role in the formation of molecules like DNA, proteins, carbohydrates, and lipids.
What macromolecule is triglyceride?
Triglycerides are macromolecules called lipids, better known as fats or oils. Triglycerides are named for the monomer components they contain. “Tri” means three, and triglycerides are built from monomers of three fatty acids bonded to a glycerol.
What is phospholipid molecule?
Phospholipids (PL) are a group of polar lipids that consist of two fatty acids, a glycerol unit and a phosphate group which is esterified to an organic molecule (X) such as choline, ethanolamine, inositol, etc.
Why phospholipid is called polar lipid?
A single phospholipid molecule has a phosphate group on one end, called the “head,” and two side-by-side chains of fatty acids that make up the lipid “tails. ” The phosphate group is negatively charged, making the head polar and hydrophilic, or “water loving.” The phosphate heads are thus attracted to the water …
Is phospholipid a lipid or carbohydrate?
The principal components of the plasma membrane are lipids (phospholipids and cholesterol), proteins, and carbohydrate groups that are attached to some of the lipids and proteins. A phospholipid is a lipid made of glycerol, two fatty acid tails, and a phosphate-linked head group.
Why are lipids also called triglycerides?
One type of lipid is called a triglyceride, an ester derived from glycerol combined with three fatty acid molecules. Figure 14.2. … Because of the long carbon chains, triglycerides are nearly nonpolar molecules and thus do not dissolve readily in polar solvents such as water.
Is triglyceride a lipid?
Triglycerides are a type of fat (lipid) found in your blood. When you eat, your body converts any calories it doesn’t need to use right away into triglycerides. The triglycerides are stored in your fat cells.
Do triglycerides have a phosphate group?
Phospholipid: Phospholipids are composed of a phosphate group attached to the glycerol backbone. Triglyceride: Triglycerides has no phosphate groups attached to the glycerol backbone.