What is the unit for first-order kinetics?

Mia Smith
Published Feb 27, 2026
What is the unit for first-order kinetics?
1/s
The units of k for a zero-order reaction are M/s, the units of k for a first-order reaction are 1/s, and the units of k for a second-order reaction are 1/(M·s).
What is the unit of 1st order reaction?
Because the units of the reaction rate are always moles per liter per second, the units of a first-order rate constant are reciprocal seconds (s−1).
What are the units of rate constant for first-order and N order reactions?
For nth order reaction, the units of rate constant = (molL−1)1−n. s−1.
What is the units of K constant?
The Coulomb constant, the electric force constant, or the electrostatic constant (denoted ke, k or K) is a proportionality constant in electrostatics equations. In SI units it is equal to 8.9875517923(14)×109 kg⋅m3⋅s−2⋅C−2….Coulomb constant.
| Value of k | Units |
|---|---|
| 10−7 | (N·s2/C2)c2 |
What is first-order kinetics?
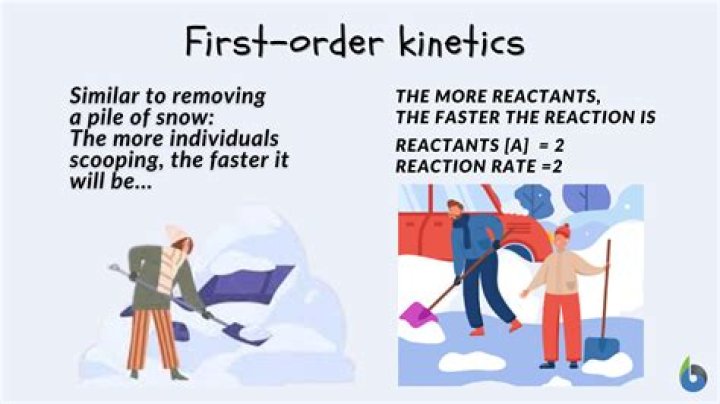
Definition. An order of chemical reaction in which the rate of the reaction depends on the concentration of only one reactant, and is proportional to the amount of the reactant.
What are the units for first-order reaction?
A second-order reaction will depend on the concentration (s) of one second-order reactant or two first-order reactants. To determine the order of a reaction with respect to each reactant, we use the method of initial rates.
What is the unit of First-Order?
k is the first-order rate constant, which has units of 1/s. The method of determining the order of a reaction is known as the method of initial rates. The overall order of a reaction is the sum of all the exponents of the concentration terms in the rate equation.
What is 1st order reaction?
Definition of first-order reaction : a chemical reaction in which the rate of reaction is directly proportional to the concentration of the reacting substance — compare order of a reaction.
What are the units of a first order rate constant?
Which of the following is the unit of rate constant for a first order reaction *?
s-1 is the unit of rate constant for a first order reaction.
What are the units of a first-order rate constant?
What are the units for third order reaction?
Some Facts About Third Order Rate Law
| Reaction Order | Units of k |
|---|---|
| Zero | mol/L/s |
| First | s-1 |
| Second | L/mol/s |
| Third | mol-1 L2 s-1 |
What are the units of the differential equation for first order kinetics?
The differential equation describing first-order kinetics is given below: The “rate” is the reaction rate (in units of molar/time) and k is the reaction rate coefficient (in units of 1/time). However, the units of k vary for non-first-order reactions.
What is the kinetics of first order reaction?
Kinetics of First order Reaction A first order reaction is one whose rate varies as 1st power of the concentration of the reactant i.e. the rate increases as number of times as the number of times the concentration of reactant is increased. Let us consider a unimolecular first order reaction represented by the general equation.
What is the rate constant of a first order reaction?
The rate constant of a first order reaction has only time unit. It has no concentration unit. This means the numerical value of k for a first order reaction is independent of the unit in which concentration is expressed. If concentration unit is changed the numerical value of k for a first order reaction will not change.
What is the 1st order unit of rate?
order. The units of rate are always M/s or Ms–1 To find the units of a rate constant for a particular rate law, simply divide the units of rate by the units of molarity in the concentration term of the rate law. Rate (Ms–1) = k[A] 1st order 1 k(units) sMs 1 M − = = −