What is the symbol for a barium ion?

Mia Smith
Published Mar 18, 2026
What is the symbol for a barium ion?
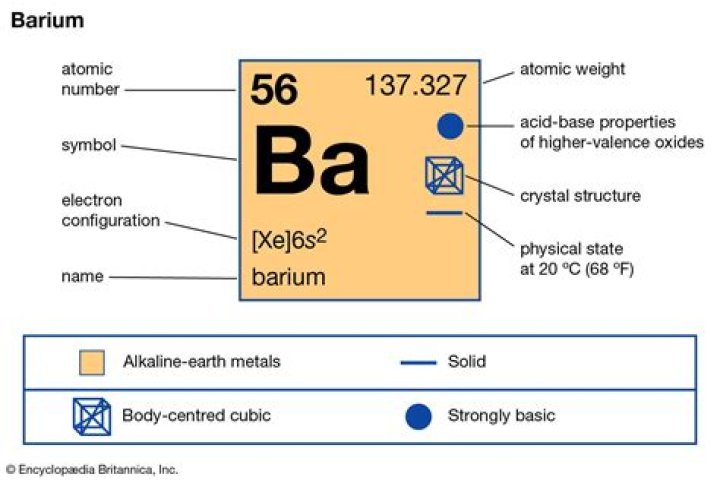
Ba
Barium(2+) is a barium cation, a divalent metal cation and a monoatomic dication. It has a role as a cofactor….4.3Related Element.
| Element Name | Barium |
|---|---|
| Element Symbol | Ba |
| Atomic Number | 56 |
How many orbitals does barium have?
Barium Atomic and Orbital Properties
| Atomic Number | 56 |
|---|---|
| Mass Number | 137 |
| Number of Neutrons | 81 |
| Shell structure (Electrons per energy level) | [2, 8, 18, 18, 8, 2] |
| Electron Configuration | [Xe] 6s2 |
What is orbital notation diagram?
Orbital diagrams are pictorial descriptions of the electrons in an atom. According to the Auf Bau Principle, each electron occupies the lowest energy orbital. The Pauli Exclusion Principle says that only two electrons can fit into an single orbital.
What is the chemical symbol of barium sulphate?
BaSO4
Barium sulfate/Formula
Is barium a metal or metalloid?
alkaline metalloid
Barium is a naturally occurring alkaline metalloid element with atomic symbol Ba, atomic number 56, and atomic weight 137 that is only found in combination with other elements, typically barite (barium sulfate) and witherite (barium carbonate), or chemicals.
How do you write orbitals?
Each orbital is written in sequence, with the number of electrons in each orbital written in superscript to the right of the orbital name. The final electron configuration is a single string of orbital names and superscripts. For example, here is a simple electron configuration: 1s2 2s2 2p6.
How do you write orbital spin?
This spin can be denoted by an arrow pointing up, which is +1/2, or an arrow pointing down, which is -1/2. Because electrons of the same spin cancel each other out, the one unpaired electron in the atom will determine the spin.
Which set of orbitals is filled last in barium?
The first two groups (columns) of the periodic table represent the ‘s’ orbital group. This means that the s,p,d,f electron configuration for Barium must end with 6s2 .
How do you find the orbital diagram?
In an orbital filling diagram, the individual orbitals are shown as circles (or squares) and orbitals within a sublevel are drawn next to each other horizontally. Each sublevel is labeled by its principal energy level and sublevel. Electrons are indicated by arrows inside the circles.
What is the symbol of barium chloride?
BaCl2
Barium chloride/Formula
How do you make an electron dot diagram for barium?
To make the electron dot diagram you put the electron symbol and put a dot on one of the sides for. Electron dot diagram for barium. The lewis structure is fbaf just draw the remaining electrons around the fluorine atoms there is a total of 16 electrons in this compound.
What is the atomic number of barium?
Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of barium (atomic number: 56), an isotope of this .Barium is an alkaline earth metal. This means that it is a group 2 element.
What is the electron configuration for nitrogen in barium?
Barium’s atomic number is 56; this means that it has 56 protons in its nucleus and also puts it as a period 6 element. In writing the electron configuration for nitrogen the first two electrons will go in the 1s orbital.
How many dots are in the Lewis structure of 133 barium chloride?
The lewis structure of the ion of chlorine has 8 dots and a 1 charge. It will gain one electron to give it a complete octet in the valence shell to form the 1 ion. Therefore the lewis structure of the element has 7 dots. The localization of 133 barium chloride in pigmented cba and albino nmri mice was reported.