What is the structure of 2/3-Dibromobutane?

Mia Smith
Published Mar 06, 2026
What is the structure of 2/3-Dibromobutane?
4.3.1FTIR Spectra
| Instrument Name | Bruker Tensor 27 FT-IR |
|---|---|
| Technique | Neat |
| Source of Spectrum | Bio-Rad Laboratories, Inc. |
| Source of Sample | Alfa Aesar, Thermo Fisher Scientific |
| Catalog Number | A10219 |
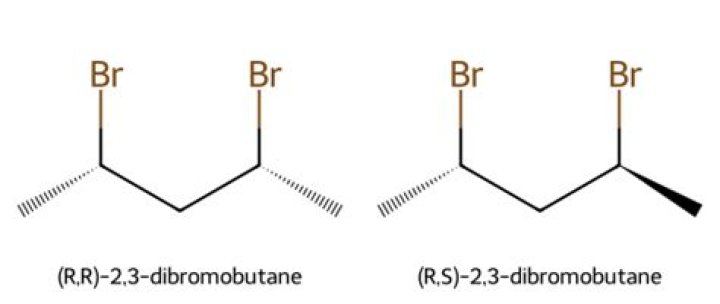
Does 2 3-Dibromobutane have a meso form?
Because it has a meso form, 2,3-dibromobutane has only 3 unique stereoisomers.
What product’s is are formed in the Debromination of Meso 2 3-Dibromobutane?
trans-2-butene
meso- 2, 3-Dibromobutane on debromination gives trans-2-butene.
Is C4H8Br2 chiral?
Chiral compounds are the unsymmetrical compound having no plane of symmetry and center of symmetry. C4H8Br2 C 4 H 8 B r 2 has a total of nine isomers; two of them contain a chiral center, one contains a chiral center but is not chiral, and six has no chiral center.
What is meso Dibromobutane?
Meso dibromobutene is the bromination product of trans-2 butene so after debromination the same product is obtained.
How many stereoisomers can 2 3 Dibromopentane have?
four stereoisomers
The four stereoisomers of 2,3-dibromopentane.
How many chiral centers does 2/3-Dibromobutane have?
Two chiral carbons
2,3-Dibromobutane Two chiral carbons, but an internal plane of symmetry.
How many pairs of enantiomers of 2/3-Dibromobutane are there?
two pairs
On the other hand, 2,3-dibromopentane has two non-equivalent stereogenic centers and there are four stereoisomers, consisting of two pairs of enantiomers.
What is obtained by Debromination of meso Dibromobutane?
meso-2 , 3- Dibromobutane on debromination gives trans-2-butene.
How many structural isomers does c4h8br2?
9 structural isomers
There are 9 structural isomers possible for this C4H8Br2
What is the name of c4h8br2?
1,2-DIBROMOBUTANE
1,2-Dibromobutane
| PubChem CID | 10792 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | C4H8Br2 |
| Synonyms | 1,2-DIBROMOBUTANE 533-98-2 Butane, 1,2-dibromo- alpha-Butylene dibromide .alpha.-Butylene dibromide More… |
Which one of the following compounds is capable of existing in a meso form?
Meso compounds are optically inactive due to internal compensation. Out of the given compounds only 2, 4-dibromopentane have a plane of symmetry, so it is a meso compound.