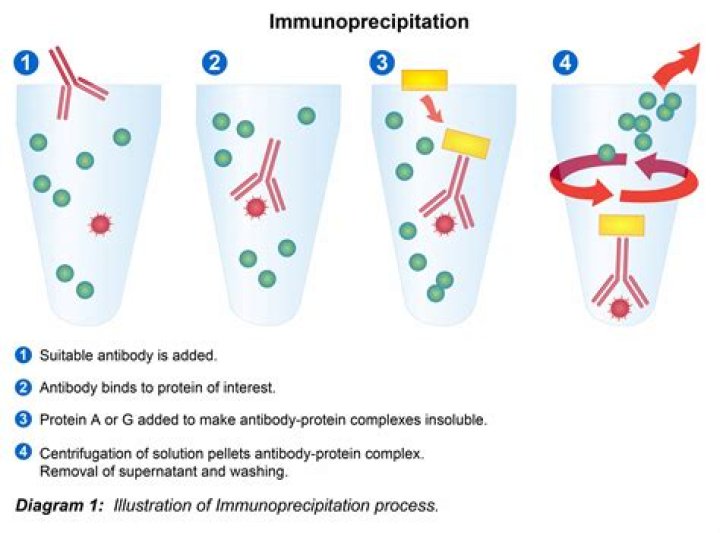
What is the principle of immunoprecipitation?

Daniel Johnson
Published Feb 24, 2026
What is the principle of immunoprecipitation?
IP is a technique that is based on the principle of antibody-antigen interaction to enrich or isolate a protein from biological samples to study its identity, structure, expression and post-translational modifications.
What is the meaning of immunoprecipitation?
Immunoprecipitation (IP) is defined as the isolation of an antigen using a specific antibody coupled (covalently or noncovalently) to a sedimentable matrix.
What is the difference between IP and co-IP?
Difference between IP and co-IP is the focus of the experiment. IP is focused on the primary target, which binds the antibody. Whereas, Co-IP targets the secondary targets, which interacts with the primary proteins, instead of antibody.
How is co-immunoprecipitation different from immunoprecipitation?
In immunoprecipitation (IP), an antibody is used to purify its specific target, or antigen from a mixture. In co-immunoprecipitation (Co-IP), an antibody is used to purify its target antigen, along with its binding partners, from a mixed sample.
Why protein A is used in immunoprecipitation?
Immunoprecipitation (IP) is the technique of precipitating a protein antigen out of solution using an antibody that specifically binds to that particular protein. This process can be used to isolate and concentrate a particular protein from a sample containing many thousands of different proteins.
What is the difference between Western blot and immunoprecipitation?
Immunoprecipitation involves using antibodies and agarose beads to isolate a target protein from a solution, while western blotting (also known as immunoblotting) uses gel electrophoresis and an antibody probe to analyze proteins.
Why do we do immunoprecipitation?
What is the purpose of co-immunoprecipitation?
Co-immunoprecipitation (co-IP) is a popular technique to identify physiologically relevant protein–protein interactions by using target protein-specific antibodies to indirectly capture proteins that are bound to a specific target protein.
What are advantages of immunoprecipitation?
Immunoprecipitation has an advantage in that the antigens are allowed to react with the antibodies in their native conformation prior to their subsequent separation and quantification. A further advantage is that a protein at a very low concentration can be concentrated from the relatively large volume of 1–2 mL.
What is immunoprecipitation western blot?
Who discovered immunoprecipitation?
In 1984 John T. Lis and David Gilmour, at the time a graduate student in the Lis lab, used UV irradiation, a zero-length protein-nucleic acid crosslinking agent, to covalently cross-link proteins bound to DNA in living bacterial cells.
How do you analyze immunoprecipitation?
Analysis of the immunoprecipitate is usually by electrophoresis although other techniques can be used. The choice of immobilized antibody binding protein depends upon the species that the antibody was raised in.