What is the pKa of alkene?

Christopher Anderson
Published Mar 11, 2026
What is the pKa of alkene?
Alkenes and alkanes, which are not acidic at all, have pKa values above 30.
What is the pKa of an alkyne?
alkanes (sp3 C-H), pKa ~ 62. alkenes (sp2 C-H), pKa ~ 45. alkynes (sp C-H), pKa ~ 26.
Which compound is expected to have highest pKa?
The phenol and ethanol both are alcohols. Out of these phenols are more acidic than phenols. So, they will be least acidic and thus will have the highest pKa value.
What is strongest acidic pKa?
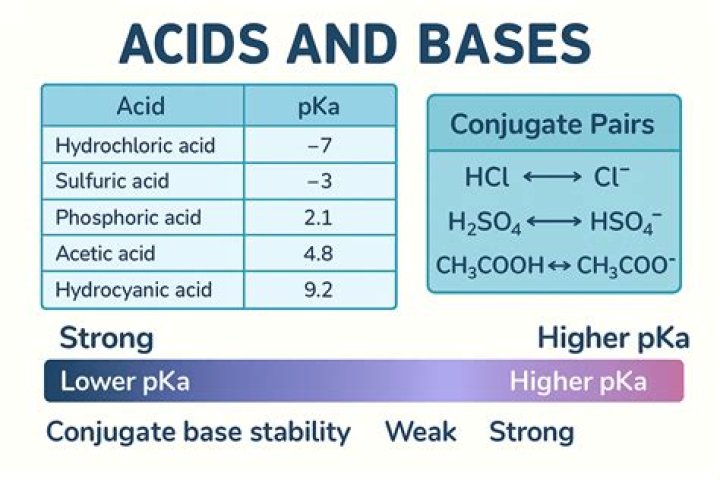
Strong acids are defined by their pKa. The acid must be stronger in aqueous solution than a hydronium ion, so its pKa must be lower than that of a hydronium ion. Therefore, strong acids have a pKa of <-174. Strong acids can be organic or inorganic.
How do you find the pKa order?
Explanation: In general the higher the acid, the higher the value of Ka and the lower the pKa=−logKa . The increasing order of acidity will reflect the decreasing order of PKa then. This order is based on the stability of the corresponding conjugate bases of the mentioned acids.
Why Terminal alkynes have a pKa of 26?
Terminal alkynes are unusual for simple hydrocarbons in that they can be deprotonated (pKa = 26) using an appropriate base (typically NaNH2, pKa = 36) to generate a carbanion (i.e. a carbon atom bearing a negative charge). This carbanion can be used as a C centered nucleophile.
Which of the following has the lowest pKa value?
Since ClCH2COOH has only one α−Cl atom and Cl2CHCOOH has two α−Cl atoms; therefore, CHCl2COOH is a strongest acid among all and has lowest pKa value.
Which of the following is the most acidic?
SO3 is the most acidic because SO3 is a most non-metallic oxide and has maximum oxidation number than all other compounds.
Is CH3COOH a weak or strong acid?
A weak acid (e.g. CH3COOH) is in equilibrium with its ions in water and its conjugate (CH3COO–, a weak base) is also in equilibrium in water.
Is HCN a strong acid?
A strong acid is an acid which is completely ionized in an aqueous solution. Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water….Strong and Weak Acids and Acid Ionization Constant.
| Acid | Conjugate Base |
|---|---|
| HCN (hydrocyanic acid) (weakest) | CN− (cyanide ion) (strongest) |
What is the pKa of an alkene?
An alkane has a pKa of roughly 50. Alkanes are very poor Bronsted bases, too: [math]CH_5^+[/math] has a pKa somewhere in the vicinity of -20. An alkene is the conjugate base of a carbocation, which is a strong Bronsted acid .
What is the pKa of functional groups?
For basic functional groups, the pKa refers to the acid strength of the base’s conjugate acid. Each functional group of a molecule with multiple ionizable functional groups has its own distinct pKa. The pKa of a specific functional group is fixed and cannot be changed.
What are the uses of alkanes and alkenes?
Uses of Alkenes. Ethene is used in the preparation of solvents like ethylene glycol, dioxane, etc. Ethylene glycol is also used as an antifreeze in automobile radiators. 3. Ethene is a plant hormone which controls growth, seed germination and fruit development. Therefore, ethene is used for artificial ripening of fruits, flower maturation, etc.
What is the pKa scale?
A more convenient scale of acidity is pKa which is the negative logarithm of the Ka (pKa = -log Ka). Thus a Ka of 1010 becomes a pKa of -10, and a Ka of 10-50 becomes a pKa of 50. More generally, more negative pKa values correspond to stronger acids and more positive pKa values correspond to weaker acids.