What is the packing efficiency of Hexagonal 2d?

Daniel Rodriguez
Published Feb 21, 2026
What is the packing efficiency of Hexagonal 2d?
The packing efficiency of two dimensional hexagon unit cell shown below is se answer: 95.5% 90.7% 76.2% 68.4%
What is the packing fraction of a 2d Hexagonal unit cell?
A. π3√2.
What is packing efficiency of HCP?
Both ccp and hcp are highly efficient lattice; in terms of packing. The packing efficiency of both types of close packed structure is 74%, i.e. 74% of the space in hcp and ccp is filled. The hcp and ccp structure are equally efficient; in terms of packing.
How do you calculate packing efficiency of 2d?
$r$ is the radius of the sphere. Thus, the percentage packing efficiency is $0.7854 \times 100\% = 78.54\% $. Thus, the packing efficiency of a two-dimensional square unit cell shown is $78.57\% $. Thus, the correct option is (D) $78.54\% $.
How do you calculate packing efficiency in HCP?
In the same way, the relation between the radius ‘r’ and edge length of unit cell ‘a’ is r = 2a and the number of atoms is 6 in the HCP lattice. Packing efficiency can be written as below, Packing efficiency = Volume occupied by 6 spheres ×100 / Total volume of unit cells.
How do you solve hexagonal packing?
The area of the hexagon can be found by splitting it into six equilateral triangles and the total area is 6\times (1/\sqrt{3}) \times 1 = 2\sqrt{3} square units. To get the proportion of the plane covered by the circles we must divide by pi by 2\sqrt{3} to get 0.90689\ldots or 90.7\% to 3 significant figures.
What is efficiency in packing?
The packing efficiency is the fraction of the crystal (or unit cell) actually occupied by the atoms. It must always be less than 100% because it is impossible to pack spheres (atoms are usually spherical) without having some empty space between them.
How do you calculate hexagonal close packing?
The central atom in layer B is surrounded by 6 atoms from layer B itself, while it is also surrounded by 3 atoms each from top and bottom layers. Hence the total number of surrounding atoms is 6+3+3 = 12, which gives us a coordination number of 12 for hcp structure. which approximately translates to 0.74048.
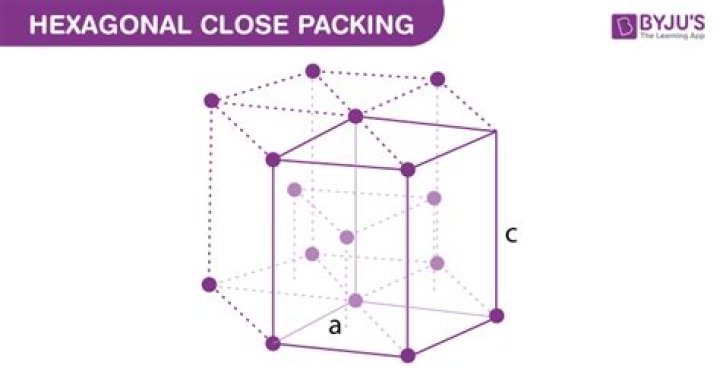
What is meant by hexagonal close packing in three dimensions?
Hexagonal Closest Packing: Here, the alternating layers fill the distance between each other. In one layer, spheres align to fit into the holes of the previous layer. There is the same alignment for the first and the third line. So we call this sort of ABA type.
What is the coordination number of 2d hexagonal close packing?
Easiest explanation: In a two-dimensional hexagonal close packed structure, each sphere is in direct contact with 6 of its adjacent spheres. Hence, it has coordination number 6.
What is the formula of packing efficiency?
Packing efficiency = Volume occupied by 6 spheres ×100 / Total volume of unit cells. Examples are Magnesium, Titanium, Beryllium etc. In body-centered cubic structures, the three atoms are arranged diagonally.
What is the packing efficiency of HCP and CCP?
Both ccp and hcp are highly efficient lattice; in terms of packing. The packing efficiency of both types of close packed structure is 74%, i.e. 74% of the space in hcp and ccp is filled.
What is 2D hexagonal close pack?
2D-Hexagonal close pack is obtained by placing 1D-One layer over constriction of another 1-D layer. Co-ordination number of 2-D Hexagonal close pack is 6. Void: The empty space in close pack is called void. Square void is in 2D-square close pack; Triangular void in 2D-Hexagonal close pack.
What is co-ordination number of 2-D square close pack?
Co-ordination number: Number of nearest neighbouring points around a point is called co-ordination number. 2-D close pack are of two types (a) 2 – D square close pack (b) 2 – D hexagonal close pack 2 – D. A A A ….. arrangement is called 2-D square close pack. Co-ordination number of 2-D square close pack is 4.
What is the packing efficiency of a unit cell?
Packing efficiency: It is the percentage of total space occupied by all the atoms present in unit cell. For H.C.P The distance between two tetrahedral voids in F.C.C is × length of body diagonal.
What is 3D close pack structure?
3D-close pack structures obtained by arrangement of one 2D layer over the other. In this we can get simple cubic, body centered, face centered (Cubic close pack), and Hexagonal close pack lattices. Simple cubic close pack can be obtained by placing one 2D-square close pack over the other each layer is identical.