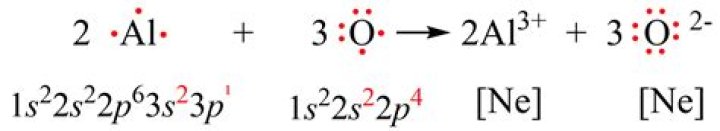
What is the name of Al2O3?

Sarah Cherry
Published Feb 10, 2026
What is the name of Al2O3?
Aluminium oxide
| IUPAC Name | dialuminum;oxygen(2-) |
|---|---|
| Alternative Names | Aluminium oxide |
| Molecular Formula | Al2O3 |
| Molar Mass | 101.96 g/mol |
| InChI | InChI=1S/2Al.3O/q2*+3;3*-2 |
Is aluminum oxide toxic to humans?
Aluminium oxides rank amongst the less toxic substances and only exhibit toxic effects in high concentrations. However, the oral intake of aluminium oxide over a long time period should be avoided as elevated aluminium levels in the blood could cause side effects on human health.
What is the coefficient for Al2O3?
The coefficient in front of Al2O3 is 2. using the chemical equation provided, identify which of the following are the coefficients of the reactants.
What is the formula for aluminium oxide?
Al₂O₃
Aluminium oxide/Formula
Is Al2O3 molecular?
Al2O3 is a solid with a three-dimensional structure and no molecule. It is not a molecule but a compound with a trigonal structure in general.
Does aluminum oxide cause Alzheimer’s?
This suspicion led to concern about exposure to aluminum through everyday sources such as pots and pans, beverage cans, antacids and antiperspirants. Since then, studies have failed to confirm any role for aluminum in causing Alzheimer’s.
Is aluminum oxide bad for skin?
Aluminum Oxide Hazards. Although aluminum oxide is certainly abrasive, it is also a skin irritant with a very questionably safety record. Aluminum oxide crystals inflame and irritate the skin, leaving it red, whereas, baking soda crystals have a natural ability to calm, tone and soothe the skin.
Is Al O2 → Al2O3 balanced?
A chemical reaction can be expressed as an equation. We will take the given equation: Al + O2 –> Al2O3. We can see that this is not balanced – there is 1 aluminum on the reactant side and 2 on the product side, and there are 2 oxygens on the reactant side and 3 on the product side.
What is the correct name for ccl4?
Tetrachloromethane
Carbon tetrachloride/IUPAC ID
Carbon tetrachloride is a manufactured chemical that does not occur naturally. It is a clear liquid with a sweet smell that can be detected at low levels. It is also called carbon chloride, methane tetrachloride, perchloromethane, tetrachloroethane, or benziform.



