What is the geometry of bef3?

Christopher Anderson
Published Mar 12, 2026
What is the geometry of bef3?
Hybridization of BF3
| Name of the Molecule | Boron Trifluoride |
|---|---|
| Molecular Formula | BF3 |
| Hybridization Type | sp2 |
| Bond Angle | 120° |
| Geometry | Trigonal Planar |
What is the shape of H3O+?
H3O+ has a molecular geometry of tetrahedral, since there are 4 regions of electron density surrounding the central O atom (3 Hydrogens and 1 lone pair of electrons). The VSEPR shape would be trigonal pyramidal.
What is the shape of AX2?
Linear electron geometry: This ball-and-stick model represents a linear compound for formula AX2. The two X atoms (in white) are 180° away from one another.
Why does water have a bent geometry?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom.
Is H3O symmetrical?
H3O+ has the same symmetry as NH3: a C3 axis, and three σv planes for a C3v molecule.
Which has a smaller bond angle H2O or H3O+?
for water will be higher due to which the bond angle will decrease. Hence H2O will have a smaller bond angle.
Is ax2 polar or non polar?
| Shape: | Linear |
|---|---|
| Steric Number: | 5 |
| Lone Pairs: | 3 |
| Polar/NonPolar: | NonPolar |
| Hybridization: | sp3d |
Why is the molecular geometry for water classified as bent and not linear?
The two hydrogen atoms and the two lone electron pairs are as far apart as possible at nearly 109obond angle. The water molecule is bent molecular geometry because the lone electron pairs, although still exerting influence on the shape, are invisible when looking at molecular geometry.
Is sof4 tetrahedral?
The molecular geometry of SOF4 is trigonal bipyramidal with asymmetric charge distribution around the central atom.
What is the bond angle of H2O?
(4) Presence of hydrogen bond. Answer: (1) An oxygen atom has 6 electrons in which two of them are bonded with a hydrogen atom leaving two lone pairs of electrons. Due to the presence of these lone pairs of electrons in H2O, the bond angle is 104.50.
Which one has the smaller bond angle?
Lone Pairs Occupy More Space than Bonding Pairs In the case of water, the two lone pairs push the bonding pairs closer together, yielding a smaller bond angle.
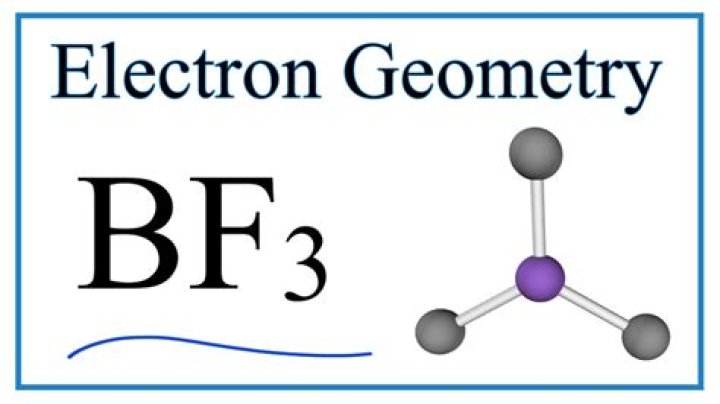
What is the shape of the molecular geometry of BF3?
Each orbital gets a single electron in an sp2 loop. The structure formed in the plane suggests that the molecular geometry of BF3 has the shape of trigonal planar (central atoms are surrounded by three-terminal atoms). This shape makes an equilateral triangle with each side making a 120-degree angle.
What is the hybridization of the molecule BF3?
As you found the molecule BF3 is sp2 hybridized (in the presence of 3 orbitals) with 1 boron atom and 3 atoms of fluorine. s-orbital is spherical. 2px and 2py orbitals are in the form of head-to-head loops.
What is the molecular geometry of water?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent ( polar bonds ).
Why is the bent shape of the water molecule critical?
The bent shape of the water molecule is critical because the polar O − H bonds do not cancel one another and the molecule as a whole is polar. The figure below illustrates the net polarity of the water molecule. The oxygen is the negative end of the molecule, while the area between the hydrogen atoms is the positive end of the molecule.