What is the difference between azacitidine and decitabine?

Mia Smith
Published Mar 07, 2026
What is the difference between azacitidine and decitabine?
Multivariate analysis revealed that compared with azacitidine treatment, decitabine treatment is significantly associated with a higher ORR (P = 0.026) and longer PFS (P = 0.037). No significant differences were observed in the incidence of grade 3 or higher haematologic adverse events in response to the two HMAs.
How long can you take azacitidine?
We strongly recommend that patients be treated with azacitidine for a minimum of 6 months, and that in patients who achieve a documented response or stable disease (sd), treatment be continued until disease progression or unacceptable toxicity occurs.
Can azacitidine cure AML?
In 2008, azacitidine was approved by the European Medicines Agency (EMA) for the treatment of AML patients with 20%–30% bone marrow (BM) blasts, older than 64 years and who are ineligible for HSCT.
What is the latest treatment for acute myeloid leukemia?
The Food and Drug Administration (FDA) recently approved two new treatments for some adult patients with acute myeloid leukemia (AML): enasidenib (Idhifa®), a drug that targets aberrant forms of the IDH2 protein; and liposomal cytarabine-daunorubicin CPX-351 (Vyxeos™), a two-drug chemotherapy combination encapsulated …
When should I take azacitidine?
Azacitidine is used to treat myelodysplastic syndrome (a group of conditions in which the bone marrow produces blood cells that are misshapen and does not produce enough healthy blood cells). Azacitidine is in a class of medications called demethylation agents.
Is azacitidine a chemotherapy?
Azacitidine is a chemotherapy drug. It is believed to work by helping your bone marrow grow normal blood cells so you will need fewer blood transfusions.
What are the side effects of azacitidine?
Azacitidine may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- nausea.
- vomiting.
- diarrhea.
- constipation.
- sores on the mouth or tongue.
- hemorrhoids.
- stomach pain or tenderness.
- heartburn.
How fast does azacitidine work?
VIDAZA is not a one-time treatment. Treatment cycles are given every 28 days for as long as your doctor recommends them. It may take several cycles (about 4 to 6 months) for your doctor to notice a difference. If you stop receiving treatment, your symptoms may return.
Can AML come back after stem cell transplant?
Disease recurrence is a devastating event after allogeneic hematopoietic stem cell transplantation as treatment for acute myeloid leukemia (AML). Median time to relapse is approximately 4 months and the majority of relapses occur within 2 years after transplant. The prognosis is usually poor.
Why is AML so hard to treat?
Generally a disease impacting older people, the average age of an AML patient is 68 at the time of diagnosis. Because it’s so aggressive, treatment for AML is considered harder on the body, especially for older patients with other health challenges.
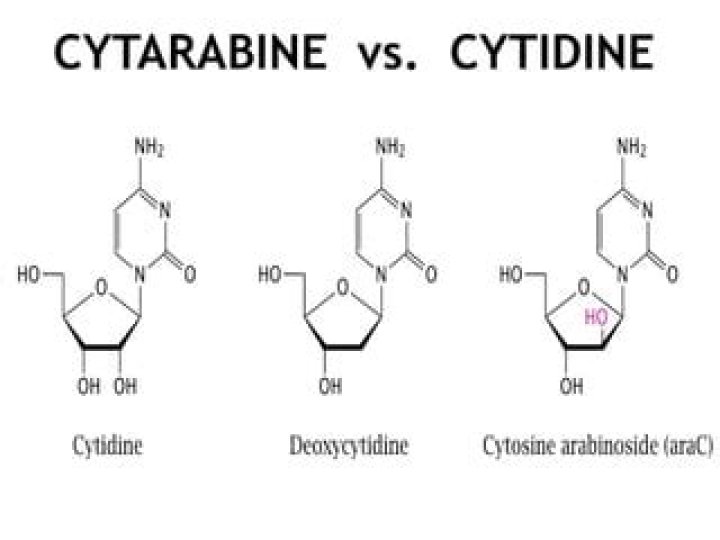
What kind of chemical is 5 Aza?
Azacitidine, sold under the brand name Vidaza among others, is a chemical analog of cytidine, a nucleoside in DNA and RNA. Azacitidine and its deoxy derivative, decitabine (also known as 5-aza-2′-deoxycytidine), are used in the treatment of myelodysplastic syndrome.
What are azacitidine (AZA) and decitabine (DAC) used for?
The cytidine nucleoside analogs azacitidine (AZA) and decitabine (DAC) are used for the treatment of patients with myelodysplastic syndromes and acute myeloid leukemia (AML).
What has been published on 5-azacitidine maintenance in AML and MDS?
An erratum has been published: Oran B, de Lima M, Garcia-Manero G, et al. A phase 3 randomized study of 5-azacitidine maintenance vs observation after transplant in high-risk AML and MDS patients.
Is decitabine or azacitidine better for myelodysplastic syndrome?
The hypomethylating agents decitabine and azacitidine have been found to improve the outcome of patients with myelodysplastic syndrome (MDS); however, the clinical choice between them is controversial. Therefore, this meta-analysis was performed to compare the efficacy, toxicity, and survival advant …
How much decitabine should patients with AML/MDS in complete remission take?
Patients with AML/MDS in complete remission (CR) after allotransplantation started decitabine between day +50 and +100. We investigated 4 decitabine doses in cohorts of 4 patients: 5, 7.5, 10, and 15 mg/m(2)/day × 5 days every 6 weeks, for a maximum 8 cycles.