What is the Colour of phenolphthalein in Na2CO3

Sarah Cherry
Published Mar 28, 2026
The phenolphthalein indicator turns pink. – The aqueous solution of sodium carbonate and sodium bicarbonate are weakly basic in nature due to hydrolysis. But sodium bicarbonate is weaker than sodium carbonate ,Therefore it does not turn the phenolphthalein indicator to pink.
What is the Colour of Na2CO3?
Sodium carbonate, Na 2CO. 3· 10H2O, (also known as Natrium Carbonate, washing soda, soda ash and soda crystals) is the inorganic compound with the formula Na2CO3 and its various hydrates. All forms are white, odourless, water-soluble salts that yield moderately alkaline solutions in water.
What is the color of phenolphthalein solution in aqueous sodium carbonate Na2CO3 solution?
when a few drops of phenolphthalein is added to an aqua solution of sodium carbonate then the colour will change to pink. Explanation: Sodium carbonate is formed by a strong base and a weak acid.
What Colour does sodium carbonate turn phenolphthalein?
Basic Procedure Put a few drops of phenolphthalein in the sodium carbonate solution. The indicator will turn pink.Which indicator is used in titration of Na2CO3?
The indicator used in the titration of sodium carbonate (weak alkali) against hydrochloric acid (strong acid) is methyl orange.
What Colour is phenolphthalein in hydrochloric acid?
Phenolphthalein is an indicator which is colourless in acidic medium and pink in basic medium. Since Hydrochloric acid is an acid,the solution will turn Colourless.
What happens when phenolphthalein is added to Na2CO3?
But sodium bicarbonate is weaker than sodium carbonate ,Therefore it does not turn the phenolphthalein indicator to pink. But sodium carbonate is relatively more basic than sodium bicarbonate and turns the solution to pink when phenolphthalein is added.
How does phenolphthalein change color?
Phenolphthalein is an indicator — a chemical which changes colour depending on whether it meets an acid or a base. It turns purple if it meets something basic, such as ammonia; it stays colourless if it meets an acid like vinegar or a neutral substance like water. … The phenolphthalein becomes colourless.Why phenolphthalein is pink in basic medium?
-Phenolphthalein is widely used as an indicator in acid-base titrations. -It turns colourless in presence of an acid and turns pink in presence of a base. … It is due to the formation of ions that the solution turns pink. Therefore, the correct answer is option (B).
Why does phenolphthalein give pink in basic medium?Phenolphthalein solution turns pink in the presence of a base. This is because the hydroxide ions react with the acidic group present and lead to the formation of ions. These ions impart color to the solution. This is how phenolphthalein solution acts as an acid-base indicator in titrations.
Article first time published onHow can you distinguish between Na2CO3 and NaHCO3?
Difference Between Sodium Carbonate and Sodium BicarbonateSodium carbonate is often referred to as soda ash or washing soda.Sodium bicarbonate is popularly called as baking soda.Sodium carbonate comes with the chemical formula Na2Co3 N a 2 C o 3 .Sodium bicarbonate formula is given as NaHCo3 N a H C o 3 .
Why phenolphthalein is used for the estimation of Na2CO3 and Naoh?
When we have both sodium carbonate and sodium hydroxide present together in a solution, a titration using phenolphthalein gives the titre (volume at the equivalence point) corresponding to sodium hydroxide plus half the carbonate and the titre obtained with methyl orange corresponds to the total alkali.
How can you distinguish Na2CO3 from NaHCO3?
When carbonate salt is reacted with MgSO4, a white salt precipited while bicarbonate salt on reacting with MgSO4, not only a white salt precipited but also a brisk effervescence occur. The gas is detected by passing it through lime water.
What is a phenolphthalein indicator?
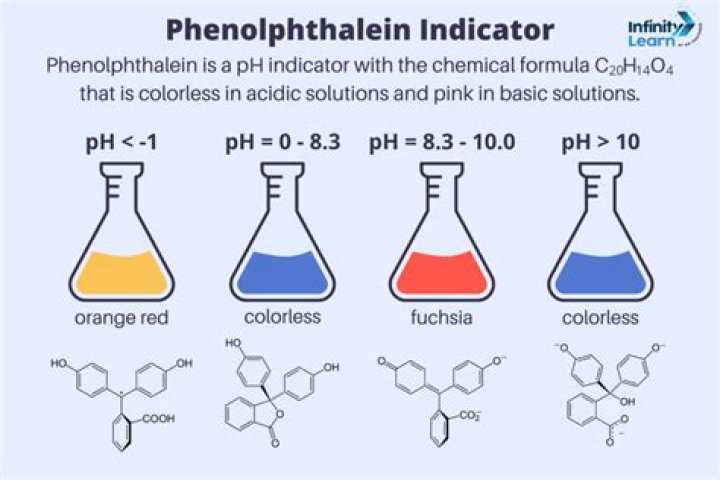
phenolphthalein, (C20H14O4), an organic compound of the phthalein family that is widely employed as an acid-base indicator. As an indicator of a solution’s pH, phenolphthalein is colourless below pH 8.5 and attains a pink to deep red hue above pH 9.0.
What is Colour of methyl orange in acid?
Methyl orange has the property to color alkaline and neutral water yellow. If the water becomes acidic, it turns red immediately.
What is the Colour change of methyl orange in acid?
Methyl orange shows red color in acidic medium and yellow color in basic medium.
What will be the Colour of phenolphthalein in lemon juice?
Phenolphthalein in neutral solution is colorless. When it is added to lemon juice it continues to remain colorless . There will be no colour change.
Why phenolphthalein Cannot show any Colour even at pH 7?
Phenolphthalein is a weak acid and is colorless in solution although its ion is pink. … Adding hydroxide ions (OH-, as found in bases) will change the phenolphthalein into its ion and turn the solution pink.
What color is Phenolphthalein in basic solution?
Phenolphthalein is often used as an indicator in acid–base titrations. For this application, it turns colorless in acidic solutions and pink in basic solutions.
What is the colour of Phenolphthalein in NaOH and HCl?
Result: Phenolphthalein gives pink colour if it is treated with basic solutions NaOH, and it remains unchanged with acidic solutions HCl.
What is the colour of Phenolphthalein in sodium hydroxide?
IndicatorColor in acidColor in basephenolphthaleincolorlesspinkmethyl orangeredyellowbromothymol blueyellowblue
Is phenolphthalein a universal indicator?
IndicatorPhenolphthaleinLow pH colourColourlessTransition pH range8.3 – 10.0High pH colourFuchsia
What is the colour of phenolphthalein in lime water?
Answer: Phenolphthalein gives pink colour with lime water. Phenolphthalein is naturally colourless but turns pink in alkaline solutions.
What is the colour of phenolphthalein in basic medium and that of methyl orange in acidic medium?
Methyl orange and phenolphthalein use as indicator , In Acidic solution Methyl orange gives red colour but in basic solution it gives yellow colour . In Acidic solution phenolphthalein is colourless but in basic it gives pink colour .
Why phenolphthalein is Colourless in acidic medium?
Detailed Solution Phenolphthalein is a weak acid. It does not dissociate in the acidic medium and remains in the unionised form. The unionised form is colourless and hence, phenolphthalein in acidic solution is Colourless.
Which solution shows pink Colour when phenolphthalein indicator is added?
Answer: CaO gives pink colour with phenolphthalein indicator in aqueous solution. An aqueous solution of CaO is alkaline in nature due to hydrolysis. The phenolphthalein indicator is colourless in acidic medium and pink coloured in alkaline medium.
Which one is more basic Na2CO3 or NaHCO3?
Answer: Na2CO3 is more basic, and NaHCO3 more acidic.
How do you identify NaHCO3?
Compare the color of the pH paper with the pH scale typically printed on the pH paper pack and assign pH of the solution accordingly. If pH is around 8, the sample is sodium bicarbonate. If pH is in the range 9.5 to 10, it is sodium carbonate.
Can phenolphthalein be used in a titration between HCl and Na2CO3?
Na2CO3 act as a weak base and HCl is a strong acid. Phenolphthalein is not a suitable indicator for titrating weak base against strong acid because the equivalence point of the titration will result in an acid salt(NaHCO3) which cannot go to completion.
Why phenolphthalein Cannot be used in determination of NaHCO3?
1. Phenolphthalein=Phenolphthalein is a compound mostly used as a ph indicator in acid-base titrations, when reacted with baking soda, phenolphthalein would change colour from colourless to pink because baking soda has a ph of 9.
What are the indicators used for the estimation of NaHCO3 and Na2CO3 in a mixture?
Estimation of Na2CO3 and NaHCO3 in a mixture: Accurately weigh about 2.0 g of the mixture and prepare a solution in distilled water in a 250 ml standard flask. Slowly titrate 25 ml of this solution against standard hydrochloric acid using phenolphthalein as indicator.



