What is the boiling point of water when salt is added?

Sarah Cherry
Published Mar 23, 2026
What is the boiling point of water when salt is added?
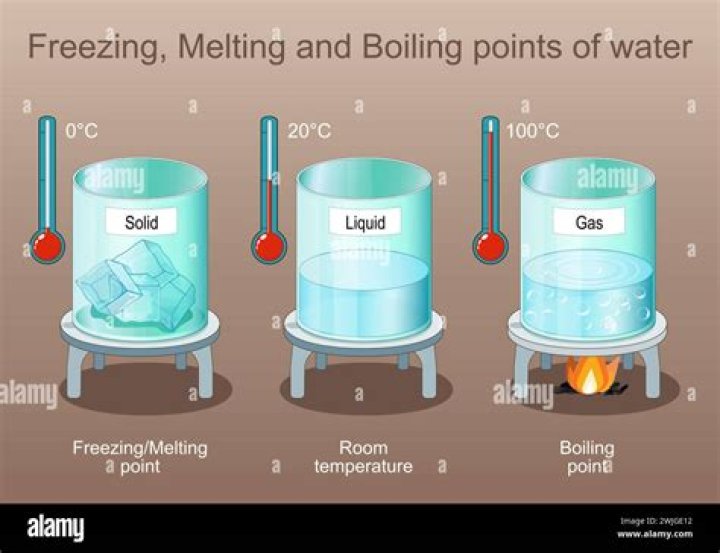
Seawater of 35 psu has a specific heat of 0.932 compared with 1.000 for pure water. Pure water freezes at 0 °C and boils at 100 °C (212 °F) under normal pressure conditions. When salt is added, the freezing point is lowered and the boiling point is raised.
How does table salt affect the boiling temperature of water?
So yes, salt increases the boiling temperature, but not by very much. If you add 20 grams of salt to five litres of water, instead of boiling at 100° C, it’ll boil at 100.04° C. So a big spoon of salt in a pot of water will increase the boiling point by four hundredths of a degree!
Does salt and water have the same boiling point?
“The temperature of saltwater will get hotter faster than that of pure water,” Giddings said. “But it still has a higher boiling point, and the mass is still greater when you add salt to the same volume of water, so this doesn’t mean that the saltwater boils faster.”
How many tablespoons of salt is in one cup of water?
Add 1 tablespoon of salt for every cup of water you used and mix until the salt is completely dissolved. For example, if you are using 1 gallon (16 cups) of water, add 16 tablespoons (1 cup) of salt.
How much does 2 cups of Epsom salt weight?
So two cups would be equal to one pound (2×8 = 16 ounce).
How much salt do I use for 2 cups of brine?
Whether you want to believe it or not, Brining Is Better!
| Water | Diamond Crystal kosher salt | Table Salt |
|---|---|---|
| 2 quarts | 1/2 cup | 1/4 cup |
| 1 gallon | 1 cup | 1/2 cup |
| 2 gallons | 2 cups | 1 cup |
| 3 gallons | 3 cups | 1-1/2 cups |
What affects the boiling point of water?
The boiling point of water, or any liquid, varies according to the surrounding atmospheric pressure. A liquid boils, or begins turning to vapor, when its internal vapor pressure equals the atmospheric pressure. Putting a liquid in a partial vacuum also will lower its boiling point.
Can boiled water go bad?
Can boiled water go bad? Boiled water can be kept in sterilized, properly sealed containers in the refrigerator for 3 days or for 24 hours if kept at room temperature out of direct sunlight.
Beaker/ Concentration of Nasal (M) Boiling Point Temperature Pure tap water 100. (00 1 tablespoon salt solution 102 (CO) tablespoon salt solution 103. 5 (CO) 3 tablespoon salt solution 105 (CO) Graph the data from Table 2 Label each axis (X and Y will NOT do). Devise a scale for each axis and label that scale (1, 2, 3…
How to boil tap water with table salt?
Kitchen stove or electric hot plate 2. One 2-cup measuring cup 3. Measuring spoon set, with one tablespoon maximum 4. Table salt 5. Tap water 6. 2 quart cooking Pot (1/2) i don’t have sorry 7. One digital thermometer Procedure (list steps in the order in which you performed them): 1. Boil one quart of tap water on stove. .
What is the boiling point of 2 h tablespoons of water?
Use your graph to determine the boiling point of a solution made with 2 h tablespoons. This use of a graph has generated a hypothesis that a 2 h tablespoon solution boils at 104 degrees C. Prepare a 2. 5 M solution and verify your hypothesis.
What is boiling point?
It depends on temperature and altitude. The simple answer to this question is that the boiling point of water is 100 C or 212 F at 1 atmosphere of pressure ( sea level ). However, the value is not a constant. The boiling point of water depends on the atmospheric pressure, which changes according to elevation.
Beaker/ Concentration of Nasal (M) Boiling Point Temperature Pure tap water 100. (00 1 tablespoon salt solution 102 (CO) tablespoon salt solution 103. 5 (CO) 3 tablespoon salt solution 105 (CO) Graph the data from Table 2 Label each axis (X and Y will NOT do). Devise a scale for each axis and label that scale (1, 2, 3…
Use your graph to determine the boiling point of a solution made with 2 h tablespoons. This use of a graph has generated a hypothesis that a 2 h tablespoon solution boils at 104 degrees C. Prepare a 2. 5 M solution and verify your hypothesis.
Kitchen stove or electric hot plate 2. One 2-cup measuring cup 3. Measuring spoon set, with one tablespoon maximum 4. Table salt 5. Tap water 6. 2 quart cooking Pot (1/2) i don’t have sorry 7. One digital thermometer Procedure (list steps in the order in which you performed them): 1. Boil one quart of tap water on stove. .
How many tablespoons of salt in a liter?
Per quart or, to again butcher numbers by rounding willy nilly, 3.6 tablespoons per liter. ( see this site for exact salt conversions to assist with salt measures salt measures conversions …) dT = 0.51 * 1 (water is 1kg per liter) * 2dT = 1.02 Which yields a rise of 1°C per 3.6 tablespoons.