What is the band gap of ZnO nanoparticles?

Sarah Cherry
Published Feb 18, 2026
What is the band gap of ZnO nanoparticles?
The optical band gap value obtained for ZnO nanoparticle at pH=7.5 is 3.63 eV.
Is ZnO direct or indirect band gap?
Zinc oxide, an important semiconductor with a direct wide band gap and large exciton binding energy, has been used for solar cells, transparent electrodes, and blue/ultraviolet light emitting devices.
What is the energy band gap of zinc oxide nanopowder?
The value of the band gap obtained for undoped ZnO is 3.31 eV and it starts increasing for 3, 5, 10, and 15 mol% samples as 3.35, 3.38, 3.40, and 3.42 eV, respectively. Fluorescence spectra of Mn-doped ZnO nanoparticles with (a) pure ZnO, (b) 3 mol%, (c) 5 mol%, (d) 10 mol%, and (e) 15 mol%.
How do you synthesize ZnO nanoparticles?
Synthesis of ZnO nanoparticles. ZnO nanoparticles were prepared by refluxing precursor zinc acetate dihydrate (0.1 M) in diethylene glycol and triethylene glycol at 180 °C and 220 °C respectively. Reaction time varied for 2 and 3 h with and without sodium acetate (0.01 M).
Is TiO2 direct band gap?
Among the 9 TiO2 polymorphs, brookite, and tridymite-structured TiO2 have direct band gap. For rutile, columbite- structured, and baddeleyite-structured TiO2, the direct band gap energies at Γ are very close to the indirect band gap energies, indicating a quasi-direct band gap character.
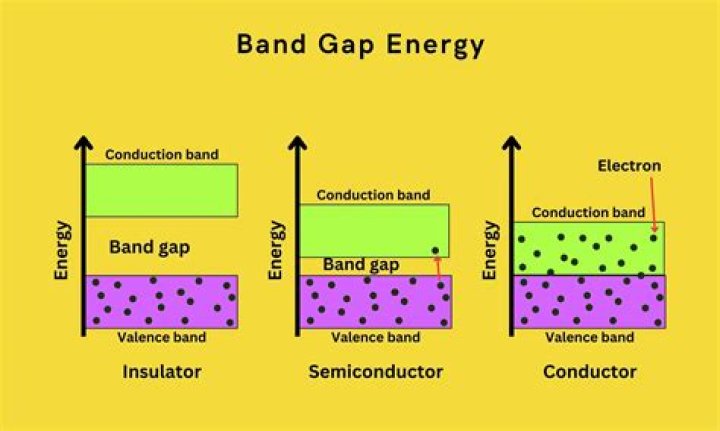
What is direct and indirect band gap?
If the k-vectors are different, the material has an “indirect gap”. The band gap is called “direct” if the crystal momentum of electrons and holes is the same in both the conduction band and the valence band; an electron can directly emit a photon. Some III-V materials are indirect bandgap as well, for example AlSb.
Is ZnO polar or nonpolar?
It is known that Zinc oxide has positive charged Zn polar surface, negative charged O polar surface, and 6 non-polar faces.
Is ZnO a semiconductor?
Although it occurs naturally as the mineral zincite, most zinc oxide is produced synthetically. ZnO is a wide-band gap semiconductor of the II-VI semiconductor group. The native doping of the semiconductor due to oxygen vacancies or zinc interstitials is n-type.
What is the band gap of TiO2?
Band gap of the TiO2 (rutile and anatase) phase is 3.2-3.35 eV. It is approximately equal to the ZnO band gap.
What is the band gap of titanium dioxide?
Even though TiO2 is an exceptional photocatalyst, its broad band gap value (3.2 eV) makes necessary the use of UV light to achieve the photoactivation.
How do you synthesize ZnO?
ZnO nanoparticles were synthesized via sol gel method using Zinc acetate dehydrate (Zn(CH3COO)2. 2H2O) as a precursor and ethanol (CH2COOH) was used as solvent, Sodium hydroxide (NaOH) and distilled water were used as medium. ZnO nanoparticles were characterized by using XRD, EDX, FESEM, and nano-particles analyser.
Why are ZnO nanoparticles synthesis?
Conclusion. In summary, ZnO NPs were synthesized by two different methods i.e., sol-gel and biosynthesis method. The green synthesis of ZnO NPs allows avoiding the toxic chemical agents that are used in the sol-gel method for the size reduction.
What is ZnO nanoparticles?
Zinc oxide nanoparticles: Synthesis, antiseptic activity and toxicity mechanism Zinc oxide (ZnO), as a material with attractive properties, has attracted great interest worldwide, particularly owing to the implementation of the synthesis of nano-sized particles.
How to prepare ZnO nanocomposites with different morphologies?
Recently, various approaches including physical, chemical and biological (“green chemistry”) have been used to prepare ZnO nanocomposites with different morphologies.
How toxic are zinc oxide nanocomposites?
Zinc oxide nanocomposites show also selective toxicity toward normal and cancerous cells, which is explained by reactive oxygen formation (ROS).