What is the balanced equation of magnesium nitride?

Daniel Rodriguez
Published Mar 11, 2026
What is the balanced equation of magnesium nitride?
The chemical formula for this reaction is Mg+N2→MgN2.
What is the formula unit for magnesium nitride?
Magnesium nitrate | Mg(NO3)2 – PubChem.
What is the formula of magnesium nitrate?
Mg(NO3)2
Magnesium nitrate/Formula
What is the formula of nitride?
N3-
The nitride ion’s formula is N3-. Nitrogen atoms need three electrons to fill its outer valence shell, which is 2p. The nucleus of a nitrogen atom…
What is the formula of sodium nitride?
Na3N
Sodium nitride/Formula
What is the formula for nitride?
N-3
The formula for nitride Ion is N-3.
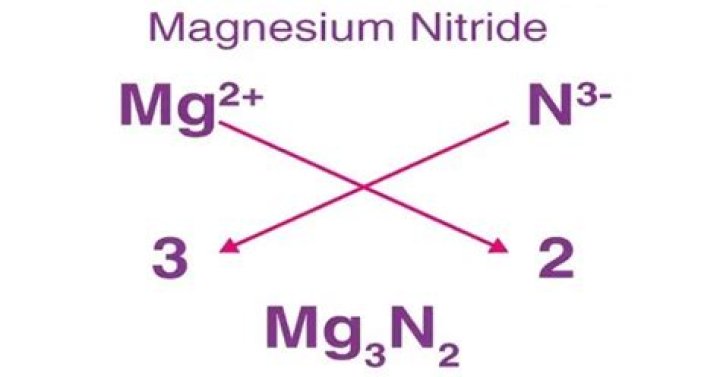
Why is magnesium nitride Mg3N2?
Problem: Magnesium nitride (Mg3N2) is formed by the reaction of magnesium metal (Mg) with nitrogen gas (N2).
What is mgn2?
Magnesium nitride reacts with water to produce magnesium hydroxide and ammonia gas, as do many metal nitrides. The Mg2N4 is composed of exotic cis-tetranitrogen N44− species with N-N bond orders close to one.
How many atoms are there in the formula of magnesium nitride?
3 atoms
It is an inorganic compound of magnesium and nitrogen consisting of 3 atoms of Magnesium and two atoms of nitrogen. The chemical or molecular formula of Magnesium Nitride Formula is Mg3N2.
What is the formula for magnesium with nitrogen?
The chemical formula for magnesium nitrite is Mg(NO2)2. The letters in the formula indicate the different elements with Mg representing magnesium, N representing nitrogen and O representing oxygen.
What is the balanced equation for heating magnesium hydroxide?
Hence, adding water to the resulting solids and heating them will produce pure magnesium oxide. Chemical Equations . The balanced chemical equation for the first part of the reaction is Mg3N2(s) + 6H2O(l) → 3Mg(OH)2(s) + 2NH3(g). With heat added, the equation for the final step of the reaction is Mg(OH)2(s) → MgO (s) + H2O(l).
What is the charge of magnesium nitrate?
Magnesium nitride is an ionic compound. Magnesium forms ions with a 2+ charge, and nitrogen forms nitride ions with a 3- charge. The overall charge of the compound must be zero.
What is magnesium and nitrogen?
MAGNESIUM NITRATE is a white crystalline solid. Produces toxic oxides of nitrogen if heated to decomposition. Used in pyrotechnics. Magnesium nitrate is the inorganic nitrate salt of magnesium.