What is saturated solution explain with example?

Daniel Rodriguez
Published Feb 13, 2026
What is saturated solution explain with example?
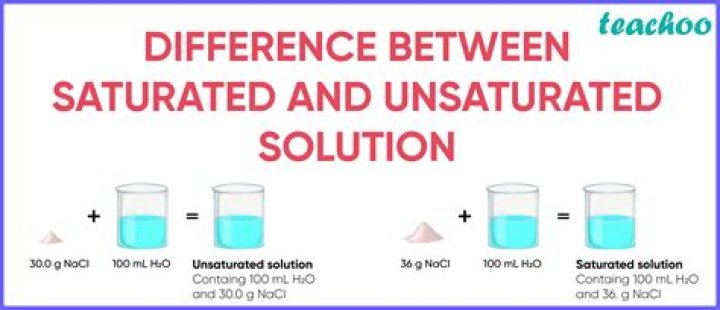
(a) Saturated Solution – A solution in which no more solute can be dissolved without raising temperature is called saturated solution. For example, 36g of salt in 100g of water. For example-Salt, Oxygen, Water.
What is the definition of saturated chemistry?
1 : being a solution that is unable to absorb or dissolve any more of a solute at a given temperature and pressure. 2 : being an organic compound having no double or triple bonds between carbon atoms.
What is unsaturated solution short answer?
Unsaturated solutions are solutions in which the amount of dissolved solute is less than the saturation point of the solvent (at that specific temperature gradient). If the amount of dissolved solute is equal to the saturation point of the solvent, the solution is called a saturated solution.
What is saturated solutions for Class 9?
-Saturated solution: A saturated solution contains the maximum amount of solute that can be dissolved in the solution at the conditions of temperature and pressure at which the solution is being prepared. If to a saturated solution, more solute is added then either a precipitate or a gas will be produced.
What is saturated and unsaturated solution with examples?
A saturated solution is one that contains the maximum amount of solute capable of being dissolved, whereas unsaturated solutions contain less than the maximum amount of solute capable of being dissolved. Sand in water is an example of an unsaturated.
What is saturation process?
Saturation is the process or state that occurs when a place or thing is filled completely with people or things, so that no more can be added. Reforms have led to the saturation of the market with goods. [ + of]
What is saturated solution Class 9th?
A saturated solution is a solution that contains the maximum amount of solute that can be dissolved under the condition at which the solution exists.
What is meant by saturated brine?
Brine is a high-concentration solution of salt in water. A saturated solution is a chemical solution containing the maximum concentration of a solute dissolved in the solvent.
What is saturated solution Brainly?
Answer: The term saturated solution is used in chemistry to define a solution in which no more solute can be dissolved in the solvent. It is understood that saturation of the solution has been achieved when any additional substance that is added results in a solid precipitate or is let off as a gas.
What is the saturated unsaturated solution?
A saturated solution is a solution that contains the maximum amount of solute that is capable of being dissolved. An unsaturated solution is a solution that contains less than the maximum amount of solute that is capable of being dissolved.
What is saturation and unsaturation?
Saturated compound : an organic molecule in which all the covalent bonds between carbon atoms are single bonds (C−C) Unsaturated compound : an organic molecule in which at least one of the covalent bonds between carbon atoms is either a double bond (C=C) or a triple bond (C≡C)



