What is keto-enol tautomerism with example?

Mia Smith
Published Mar 09, 2026
What is keto-enol tautomerism with example?
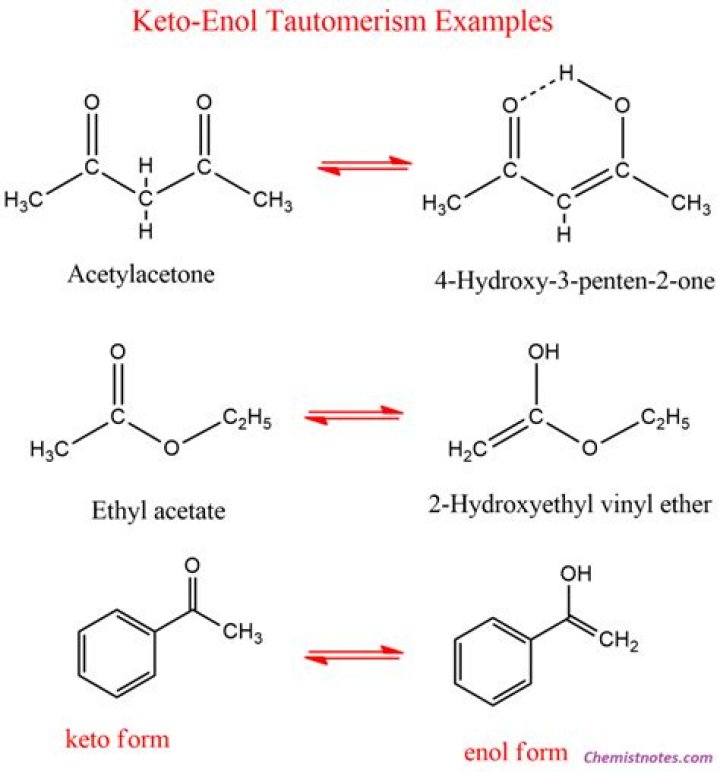
In organic chemistry, keto–enol tautomerism refers to a chemical equilibrium between a keto form (a ketone or an aldehyde) and an enol (an alcohol). The keto and enol forms are said to be tautomers of each other.
What is keto-enol isomerism?
Keto-enol tautomerism refers to a chemical equilibrium between a keto form (a ketone or an aldehyde) and an enol (an alcohol). The interconversion of the two forms involves the movement of an alpha hydrogen and the shifting of bonding electrons; hence, the isomerism qualifies as tautomerism.
Which technique is used to study keto-enol tautomerism?
Chemical Reactions Studied By Electronic Spectroscopy.
What statement is correct for keto-enol tautomerism?
Generally, the chemical equilibrium in keto-enol tautomerism is extremely thermodynamically driven. Generally at room temperature equilibrium heavily favors the existence of the keto form. If we increase the temperature a little bit more then keto converts into enol form. So, option B is also correct.
What is meant by Keto enol tautomerism explain with an example?
Tautomers are isomers of a compound that differ only in the position of the proton and electrons. Keto-enol tautomerism refers to a chemical equilibrium between the keto form (carbonyl structure containing α-hydrogen) and the enol form (a double bond adjacent to an alcohol, -C=C-OH) of a compound.
Why keto form is more stable?
In general , keto form is more stable than enol form due to greater bond energy of c=o. than c=c .
What is meant by Keto-enol tautomerism?
Keto-enol tautomerism refers to a chemical equilibrium between the keto form (carbonyl structure containing α-hydrogen) and the enol form (a double bond adjacent to an alcohol, -C=C-OH) of a compound. Its stability depends upon factors like aromaticity, hydrogen bonding, solvent, conjugation, and substitution [1-5].
Why does keto-enol tautomerism occur?
In the case of a terminal alkyne, when a single pi bond is turned into an alcohol, the terminal carbon still has a pi bond or alkene, making it an enol. This molecule will also undergo tautomerization to form a more stable aldehyde product.
What is meant by Keto-enol tautomerism explain with an example?
Which of the following gives keto-enol tautomerism?
Both I and III. hydrogen undergoes keto-enol tautomerism.
What is meant by keto-enol tautomerism?
What is keto-enol tautomerism Class 11?
The “keto-enol” tautomerism is an acid/base catalysed mechanism where the carbonyl form is initially protonated and thereby the conjugate base formed extracts proton from one of the alkyl carbon atoms while the shared electron share between the hydrogen atom and the carbon atom forms the double bond forming the enol.