What is hybridisation of NO2 plus?

Mia Smith
Published Feb 16, 2026
What is hybridisation of NO2 plus?
So, the species is tetrahedral with sp3 hybridisation. So, the species is linear with sp hybridisation.
What is hybridisation formula?
Hybridization=1/2(valency electron in central atom+no. Of atom attached to central atom by single bond+negative charge-positive charge).
What is the hybridization of nitrogen?
sp3hybridized
The nitrogen is sp3hybridized which means that it has four sp3 hybrid orbitals. Two of the sp3hybridized orbitals overlap with s orbitals from hydrogens to form the two N-H sigma bonds.
What is the hybridization of nitrogen in NO2+?
The hybridisation of atomic orbitals of nitrogen in NO2+, NO3- and NH4+are 1. sp, sp and sp respectively 2.
How do you find the hybridization of nitrogen?
How To Determine Hybridization: A Shortcut
- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
How do you find the hybridization of NO2?
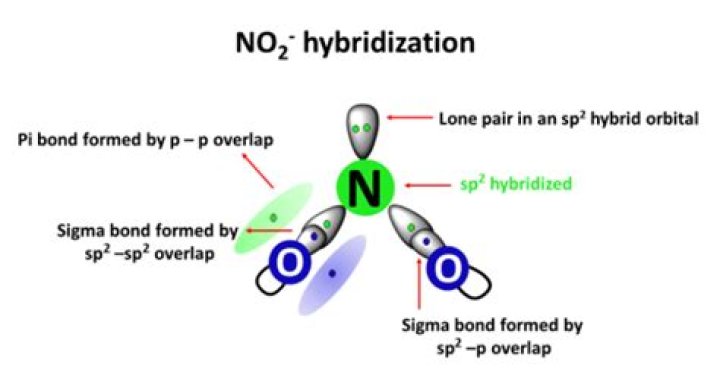
NO2 involves an sp2 type of hybridization. The most simple way to determine the hybridization of NO2 is by drawing the Lewis structure and counting the number of bonds and lone electron pairs around the nitrogen atom. You will find that in nitrogen dioxide there are 2 sigma bonds and 1 lone electron pair.
Is nitrogen a sp2?
In aniline, the nitrogen atom is planar and thus sp2 hybridized. This occurs because the lone pair of electrons on the nitrogen is now in a 2p orbital, and thus able to delocalize into the aromatic ring. The fact that aromatic amines like aniline are planar and sp2 hybridized is extremely important in biology.
What is the hybridisation of nitrogen in the following species a NO2 B NO3?
sp2, sp3 and sp respectively.
What is the hybridization of anionic part of solid cl2o6 molecule?
sp3d2.
How do you find the hybridization of no2?
What is the hybridization of nitrogen in pyridine?
sp2 hybridized
Pyridine is an example of a six-membered aromatic heterocycle and has an electronic structure similar to benzene. In the bonding picture of pyridine the five carbons and single nitrogen are all sp2 hybridized.
What is hybridization of nitrogen?
The nitrogen is sp3hybridized which means that it has four sp3 hybrid orbitals. Two of the sp3hybridized orbitals overlap with s orbitals from hydrogens to form the two N-H sigma bonds. Due to the sp3 hybridization the nitrogen has a tetrahedral geometry.