What is ground state and excited state in electron configuration

Christopher Anderson
Published Apr 04, 2026
The ground state configuration is the lowest energy, most stable arrangement. An excited state configuration is a higher energy arrangement (it requires energy input to create an excited state). Valence electrons are the electrons utilised for bonding.
What is ground and excited state?
The ground state of an electron, the energy level it normally occupies, is the state of lowest energy for that electron. … When an electron temporarily occupies an energy state greater than its ground state, it is in an excited state.
What is ground state electron configuration?
The ground state electron configuration is the arrangement of electrons around the nucleus of an atom with lower energy levels. The electrons occupying the orbitals of varying energy levels naturally falls towards the lowest energy state or ground state.
What is excited state in electron configuration?
An excited state means that (typically) the valence electron has moved from its ground state orbital (i.e. lowest available energy) to some other higher energy orbital.What is the meaning of excited state?
Definition of excited state : a state of a physical system (such as an atomic nucleus, an atom, or a molecule) that is higher in energy than the ground state.
What is the excited state of sodium?
The first excited state is the same configuration as the ground state, except for the position of one electron. As an example, sodium goes through a 3s→3p transition. This corresponds to an excitation to a first excited state that is less stable; that then leads to a relaxation back down to the ground state.
What is the difference between ground state and excited state Class 11?
The main difference between ground state and excited state is that ground state is a state where electrons in a system are in the lowest possible energy levels whereas excited state is any state of the system that has a higher energy than the ground state.
What do you mean by ground state?
Definition of ground state : the state of a physical system (as of an atomic nucleus or an atom) having the least energy of all the possible states. — called also ground level.What is the excited state electron configuration of carbon?
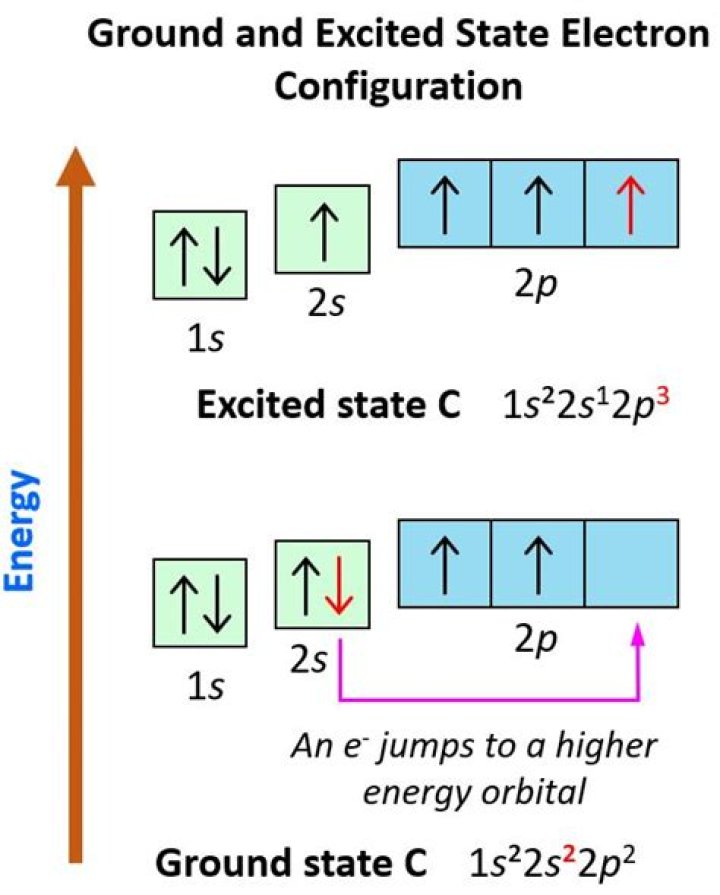
An excited state electron configuration of carbon is 1s2 2s1 2p3.
Which two elements have the same ground state electron configuration?(d) Cl and Ar have the same ground-state electron configuration.
Article first time published onWhat is excitation state physics?
excitation, in physics, the addition of a discrete amount of energy (called excitation energy) to a system—such as an atomic nucleus, an atom, or a molecule—that results in its alteration, ordinarily from the condition of lowest energy (ground state) to one of higher energy (excited state).
Which are excited states?
In quantum mechanics, an excited state of a system (such as an atom, molecule or nucleus) is any quantum state of the system that has a higher energy than the ground state (that is, more energy than the absolute minimum). Excitation is an elevation in energy level above an arbitrary baseline energy state.
When an electron is raised from the ground state to excited state?
Hence, when a hydrogen atom is raised from ground state to excited state, its potential energy increases whereas kinetic energy decreases.
What is excited state Class 11?
When energy is supplied to an atom ,an electron in the atom may jump from its normal energy level to some high energy level by absorbing a definite amount of energy. This state of atom is called excited state .
What is the energy difference between the electrons ground state and its first excited state?
The energy difference between the ground state and first excited state is 10.2 eV. If the electron absorbs a photon, the energy of the photon goes into raising the electron to an excited state. The excited states of the electron are quantized, that is, only certain energy levels are allowed.
What is the difference between hydrogen atom in the ground state and excited state?
The lowest energy state (ground state) of an atom has its electrons filling orbitals beginning from lowest energy, with 2 electrons per orbital. When energy is added to the ground state atom, an electron can be promoted to one of the higher energy orbitals. … This hydrogen molecule is in an excited state.
What is ground state of sodium?
Sodium atoms have 11 electrons and the shell structure is 2.8. 1. The ground state electron configuration of ground state gaseous neutral sodium is [Ne]. 3s1 and the term symbol is 2S1/2.
Which electron configuration represents an excited state for a potassium atom?
Which electron configuration represents an excited state for a potassium atom? An atom in an excited state has an electron configuration of 2-7-2. Identify the electron configuration of this atom in the ground state. What is the total number of valence electrons in a calcium atom in the ground state?
What is ground state carbon?
A ground-state atom is an atom in which the total energy of the electrons can not be lowered by transferring one or more electrons to different orbitals. That is, in a ground-state atom, all electrons are in the lowest possible energy levels. … Therefore, this carbon atom is a ground-state atom.
What is the ground state configuration of calcium CA?
For instance, the ground state electronic configuration of calcium (Z=20) is 1s22s22p63s23p64s2. The calcium ion (Ca2+), however, has two electrons less.
What is the ground state electron configuration of carbon quizlet?
The electron configuration for the carbon atom (C) is 1s2 2s2 2p2.
What is meant by an excited electron?
When an electron temporarily occupies an energy state greater than its ground state, it is in an excited state. An electron can become excited if it is given extra energy, such as if it absorbs a photon, or packet of light, or collides with a nearby atom or particle.
What is first excited state?
Electrons have the most potential energy when they are in an excited state. Excited state is defined as the energy state which is higher than the ground state. For the first excited state, ${\text{n = 2}}$.
Why is ground state important?
The ground state refers to an unexcited atom where the electrons are in their lowest energy levels. … The photon can tell us how many energy levels the excited level jumped. Using the ground state of electrons can also tell us the fill order of electrons in an atom.
Is Li and Na have the same ground state electron configuration?
E. No two elements have the same ground-state electron configuration.
Is electron configuration the same as ground state electron configuration?
The arrangement of electrons in the atomic orbitals of an atom is called the electron configuration. Electron configurations can be determined using a periodic table. The ground state configuration is the lowest energy, most stable arrangement.
Do different atoms have different ground states?
Atoms may occupy different energy states. … Therefore an atom can only move to a new energy level if it absorbs or emits an amount of energy that exactly corresponds to the difference between two energy levels. The lowest possible energy level that the atom can occupy is called the ground state.
What is excitation and de excitation of electrons?
atomic de-excitation: process by which an atom transfers from an excited electronic state back to the ground state electronic configuration; often occurs by emission of a photon. laser: acronym for light amplification by stimulated emission of radiation. phosphorescence: the de-excitation of a metastable state.
What is excitation potential in physics?
The amount of energy required to raise the energy level of an atom; a necessity if the atom is to radiate energy. Low excitation potential is the amount of energy, expressed in electron volts, needed to stimulate an atom to the state in which it can absorb the light of a given wavelength. …
What is an excited nucleus?
In an excited state, one or more of the protons or neutrons in a nucleus occupy a nuclear orbital of higher energy than an available nuclear orbital. These states are analogous to excited states of electrons in atoms. When excited atomic states decay, energy is released by fluorescence.