What is FCS microscopy?

Christopher Anderson
Published Feb 28, 2026
What is FCS microscopy?
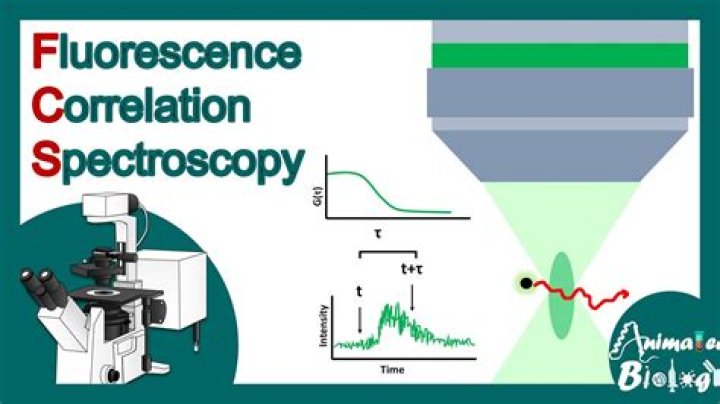
Fluorescence correlation spectroscopy (FCS) is a technique in which spontaneous fluorescence intensity fluctuations are measured in a microscopic detection volume of about 10-15 L (1 femtoliter) defined by a tightly focused laser beam.
What is FCS in biology?
Fluorescence correlation spectroscopy (FCS) is a microscopy method in which the fluorescence intensity arising from molecules within a small volume is collected over time and correlated to obtain information regarding dynamics and concentrations. From: Methods in Cell Biology, 2014.
What is technique it use FCS?
FCS is a technique basically used for spatial and temporal analysis of molecular interactions of extremely low-concentration biomolecules in solution.
What is FCS camera?
Cameras. As FCS is a technique that looks at a small number of molecules, the field of view (FOV) of a camera is an unimportant factor, with FCS detectors instead needing to focus on speed and sensitivity.
What is FRAP technique?
Fluorescence recovery after photobleaching (FRAP) is a microscopy-based method used to study the mobility of fluorescent molecules that has been employed since the mid-1970s to assess mobility of molecules in the cell membrane.
How does single molecule FRET work?
With smFRET, we utilize the natural phenomenon that occurs between 2 fluorophores, termed donor and acceptor, when they come within 1-10 nm. The emission energy of the donor is transferred to the acceptor which subsequently fluoresces when in close proximity.
Is FCS the same as FBS?
There is no difference between FBS and FCS, it is just a naming preference. So FBS and FCS are the same and both fetal, but different to BS or CS which would come from a calf already born.
Who invented fluorescence correlation spectroscopy?
Magde
Fluorescence Correlation Spectroscopy or FCS was first invented by Magde, Elson and Webb in the seventies of the last century [1], [2], [3]. Since then, it has become an indispensable tool for many applications in biology, biophysics, chemistry or physics [4], [5], [6], [7].
How do you measure fluorescence anisotropy?
It can be measured by moving the excitation polarizer to the horizontal orientation and comparing the intensities when the emission polarizer is vertically and horizontally polarized respectively. G is emission wavelength dependent. Note G in literature is defined as the inverse shown.
What is the bleached area in a FRAP?
Thanks to the development of GFP fusion proteins, a revival of FRAP applications was seen in the mid-1990s. In FRAP, fluorescent molecules in a region of interest (ROI) within the plasma membrane are bleached by a high-intensity laser source.
What is FRET microscopy?
The technique of fluorescence resonance energy transfer (more commonly referred to by the acronym FRET), when applied to optical microscopy, permits determination of the approach between two molecules within several nanometers (see Figure 1), a distance sufficiently close for molecular interactions to occur.
What is the Forster distance?
The Förster distance represents the molecular separation at which energy transfer is 50-percent efficient. For measurable FRET to occur, several requirements must be met. Among these is a strong dependence on the physical distance between the donor and acceptor fluorophores, which is limited to only a few nanometers.



