What is chiral and a chiral molecule?

Rachel Hickman
Published Mar 15, 2026
What is chiral and a chiral molecule?
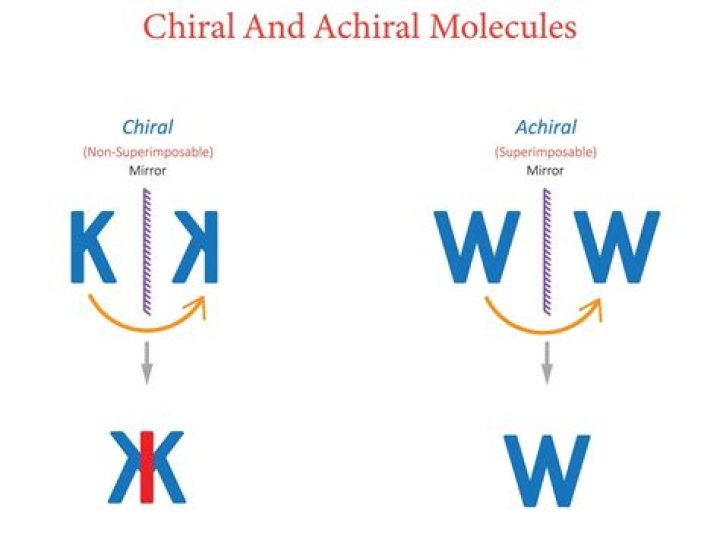
The term chiral, from the Greek work for ‘hand’, refers to anything which cannot be superimposed on its own mirror image. Chiral molecules contain one or more chiral centers, which are almost always tetrahedral (sp3-hybridized) carbons with four different substituents. …
What is the difference between chiral and a chiral?
A chiral is an object that is not identically superimposable with a mirror image of itself. An achiral is an object that is identically superimposable with a mirror image of itself.
What is a chiral substance?
In chemistry, a molecule or ion is called chiral (/kaɪˈræl/) if it cannot be superposed on its mirror image by any combination of rotations, translations, and some conformational changes. The two enantiomers have the same chemical properties, except when reacting with other chiral compounds.
What is a chiral atom?
Chiral Center, chiral atom, chirality center, or center of chirality is a tetrahedral atom in a molecule bearing four different ligands, with lone pairs, if any, treated as ligands. If a chiral center is a carbon atom, it can also be called an asymmetric carbon atom.
What are chiral molecules with examples?
One example of a chiral molecule is glucose, that naturally only occurs in the so-called right-hand variety, called D-glucose or dextrose (dexter=Latin for right). It is possible to make L-glucose (its mirror-image) by chemical synthesis.
What is chiral molecule explain with example?
A chiral molecule is a molecule that is not superimposable on its mirror image. eg. 1: Molecule 1 is not superimposable on its mirror image and, therefore, is chiral. Molecule 3 is superimposable on its mirror image and, therefore, is achiral.
What is chiral molecule class 12?
Chirality is defined as an object or a compound which is asymmetric and cannot be superimposed over its mirror image (for example, our both palms) is known as ‘chiral’ or ‘stereocenter’ and this property is known as “chirality”.
What is chirality explain with example?
In mathematics, a figure is chiral (and said to have chirality) if it cannot be mapped to its mirror image by rotations and translations alone. For example, a right shoe is different from a left shoe, and clockwise is different from anticlockwise.
What is meant by a chiral molecule?
Chirality is an important geometric property relating to a molecule’s symmetry. A chiral molecule is non-superimposable with its mirror image, and has a “handedness” (think of shoes, which specifically go with a right or left foot).
What is chirality explain?
Chirality, or handedness, means that an object or molecule cannot be superimposed on its mirror image by any translations or rotations [313]. Achiral (not chiral) objects are those objects that are identical to their mirror image.
What is the example of chiral molecule?
What is chirality define with examples?
How do you know if a molecule is chiral?
There is a simple way to tell if a molecule is chiral. It applies to most cases, but beware that some molecules which are chiral are not covered by this simple rule. The rule is this: A molecule that has a carbon atom connected to four different atoms (or groups of atoms) will be chiral.
What does it mean to have a chiral molecule?
A chiral molecule is a type of molecule that has a non-superposable mirror image. The feature that is most often the cause of chirality in molecules is the presence of an asymmetric carbon atom. The term chiral in general is used to describe an object that is not superposable on its mirror image.
How is the chirality of a molecule determined?
How to test for chirality (a) (Most reliable) Check to see if the molecule has a pair of non-superimposable mirror image forms. (b) (Nearly always works) Look for a mirror plane; if no mirror plane, the molecule is nearly always chiral (c) Look for chiral centers – tetrahedral atoms (usually carbon) with four different groups attached.
What is the difference between chiral and enantiomers?
Chiral is only describing what a single atom looks like; that it has four distinct groups attached. So, the main difference between chiral and enantiomers is what they are describing, chiral describes the atom , while enantiomers describes the molecules’ comparison.



