What is ANDA and NDA?

John Castro
Published Mar 18, 2026
What is ANDA and NDA?
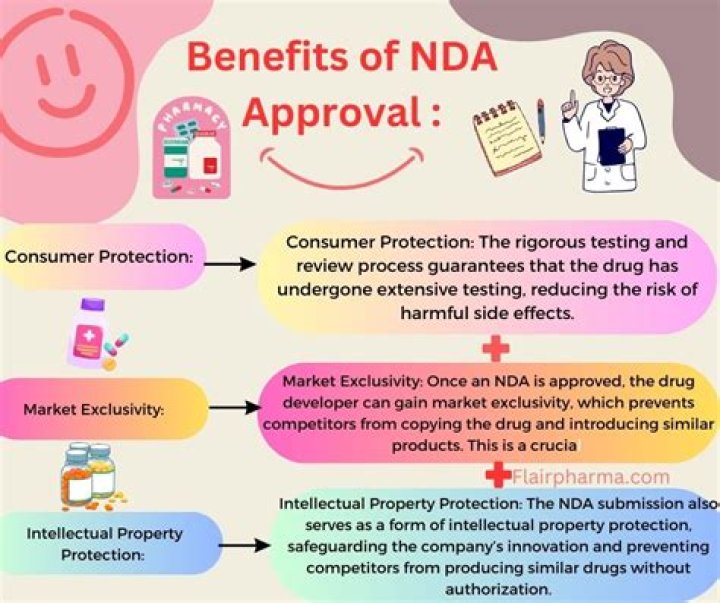
An Abbreviated New Drug Application (ANDA) contains data which is submitted to the FDA for the review and potential approval of a generic drug product whereas a New Drug Application (NDA) is the application through which sponsors formally propose the approval of a new pharmaceutical drug.
What is included in an ANDA?
ANDA Specifications An ANDA lists the new drug’s established name, trade name (if any), chemical name, dosage form(s), and strength(s), route of administration, and proposed use. The ANDA asks for the name of the listed drug product to which the proposed generic is an equivalent.
When can you file an ANDA?
Absent an NCE exclusivity, which blocks the ANDA filing (not merely the approval), an ANDA may be filed at any time after NDA approval.
Are ANDAs public?
In its recently-published final rule on Abbreviated New Drug Applications, the US Food and Drug Administration has stated that it will continue with its policy of making ANDA suitability petitions available to the public. The FDA agreed with the latter view. …
What is the purpose of ANDA?
An abbreviated new drug application (ANDA) contains data which is submitted to FDA for the review and potential approval of a generic drug product. Once approved, an applicant may manufacture and market the generic drug product to provide a safe, effective, lower cost alternative to the brand-name drug it references.
What is IND and Anda?
In short, IND application is to declare to the authorities for clinical trials; NDA application is to report to the authorities for drug registration and marketing; and ANDA application refers to the application for registration and marketing of generic drugs.
Which of the federal law includes information about ANDA submission?
This guidance is intended to assist applicants in preparing abbreviated new drug applications (ANDAs) for submission to FDA under section 505(j) of the Federal Food, Drug, and Cosmetic Act (FD&C Act) (21 U.S.C. 355(j)).
What criteria must a generic drug meet to gain FDA approval?
The generic drug needs to show that it is the same type of product (such as a tablet or an injectable) and uses the same time release technology (such as immediate-release, meaning for immediate effect of the drug, or extended-release, meaning one that is intended to slowly release the active ingredient over time).
Which is the top pharmaceutical company by revenue in the world today?
Top 25 pharma companies by global sales
| # | Company | Growth ($m) |
|---|---|---|
| 1 | Pfizer Inc | 2161 |
| 2 | Novartis AG | 4409 |
| 3 | F. Hoffmann-La Roche Ltd | 2404 |
| 4 | Johnson & Johnson | 3767 |
Which federal laws includes information about ANDA submissions?
What is DMF in pharma?
A Drug Master File (DMF) is a submission to the Food and Drug Administration (FDA) that may be used to provide confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of one or more human drugs.
What is the difference between Ind NDA and ANDA?



