What happens when cyclohexene reacts with bromine?

Michael Henderson
Published Mar 18, 2026
What happens when cyclohexene reacts with bromine?
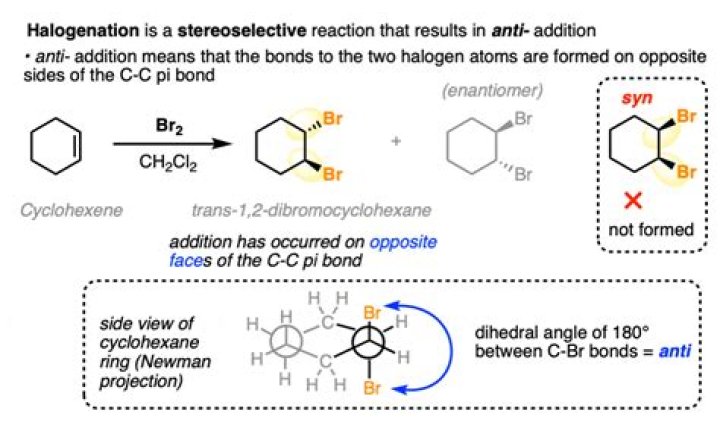
Cyclohexene reacts with bromine in the same way and under the same conditions as any other alkene. 1,2-dibromocyclohexane is formed. The reaction is an example of electrophilic addition.
What is the reaction mechanism for bromination?
Reaction Overview: The alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as Cl2 or Br2 is added to a molecule after breaking the carbon to carbon double bond. The halides add to neighboring carbons from opposite faces of the molecule.
What is the reaction mechanism for the bromination of alkene what intermediates are involved?
So in this reaction, the alkene acts as a nucleophile, attacking the electrophilic bromine, giving rise to a 3-membered ring intermediate. This is then attacked from the back side [similar to the backside attack in the SN2] at the carbon best able to stabilize positive charge, to give the trans product.
What happens when cyclohexene reacts with HBr?
Alkenes react with hydrogen bromide in the cold. The double bond breaks and a hydrogen atom ends up attached to one of the carbons and a bromine atom to the other. In the case of ethene, bromoethane is formed. With cyclohexene you get bromocyclohexane.
What is the type of reaction between cyclohexane and bromine in dichloromethane?
The only reaction that may occur between cyclohexane and bromine is free-radical substitution. Ultraviolet light radiation is required for this reaction to occur. No reaction is shown until the mixture is not treated with ultraviolet radiation.
What does cyclohexane react with?
Reactions with cyclohexane Pure cyclohexane in itself is rather unreactive, being a non-polar, hydrophobic hydrocarbon. It can react with very strong acids such as the superacid system HF + SbF5 which will cause forced protonation and “hydrocarbon cracking”.
What type of reaction is bromination?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution.
Is bromination anti or syn?
Bromination of Alkenes Gives anti Products Although we saw that several key reactions of alkenes were consistent with this mechanism, it isn’t the case for all.
Why does alkenes undergo electrophilic addition explain with reaction mechanism and suitable examples?
Why do alkenes undergo electrophilic addition reactions? Alkenes are doubly bound and sp2 hybridized, which can be donated to an electrophile, such as electrophilic addition, by the electrons in the side-to-side overlap of p orbitals that allows the pi bond.
What product would you expect from reaction of cyclohexene with HBr with HCl?
Bromocyclohexane and chlorocyclohexane are formed.
What is the product of the reaction between cyclohexane and one mole of bromine water Br2 in the presence of UV light?
Cyclohexene reacts with bromine in the same way and under the same conditions as any other alkene. 1,2-dibromocyclohexane is formed.
Does cyclohexane undergo addition reaction?
This sequence demonstrates that alkanes and benzene do not react with bromine in the absence of a catalyst. Alkenes like cyclohexene undergo addition reactions with bromine to produce dibromoalkanes.
What happens when bromine is added to cyclohexene?
If bromine water is put into a glass of cycloHEXANE, the solution will turn the yellowish/orange colour of the bromine water, as no reaction takes place, whereas if it is put in cycloHEXENE, the solution will go colourless, as the bromine bonds with the cyclohexene. Read about it: Is Potassium a polyatomic ion?
What happens when Cyclohexene is polarized?
On approaching the electron dense area of the p bond of cyclohexene, the bromine molecule becomes polarized. The electron density of the bromine is shifted, so that one bromine is partially positive and the other is partially negative charged.
Is cyclohexene an alkene or benzene?
Cyclohexene is a typical alkene, and benzene and anisole are aromatic compounds. What is the formula of bromine water? Bromine water, also called Bromide bromate solution or Bromine solution with the chemical formula Br2. The molecular weight of bromine water is 159.81 and the density is 1.307 g/mL.
What is formed when ethene reacts with bromine?
In the case of the reaction with ethene, 1,2-dibromoethane is formed. This decolourisation of bromine is often used as a test for a carbon-carbon double bond. If an aqueous solution of bromine is used (“bromine water”), you get a mixture of products.