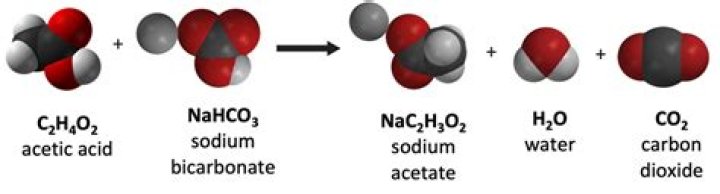
What happens when bicarbonate reacts with water?

Daniel Johnson
Published Feb 11, 2026
What happens when bicarbonate reacts with water?
When baking soda or sodium Bicarbonate (NaHCo3) reacts with water resulting in release of carbonic acid. The reaction is exothermic that means it gets the heat out. It will dissolve in water making it slightly more basic since sodium bi carbonate is alkaline in nature.
Does bicarbonate dissociate in water?
Sodium bicarbonate (NaHCO3), also known as baking soda or sodium hydrogen carbonate, is a white powder that readily dissolves in water to produce sodium (Na+) ions and bicarbonate (HCO3) ions. In the presence of acids, these ions create carbon dioxide gas (CO2) and water.
Does bicarbonate ion react with water?
When carbon dioxide mixes with the water it is partially converted into carbonic acid, hydrogen ions (H+), bicarbonate (HCO3–), and carbonate ions (CO32–)….When carbonate formation loses equilibrium.
| Accept | |
|---|---|
| Cookie Laufzeit | 2 months |
What is the chemical formula of bicarbonate?
HCO3-
Bicarbonate/Formula
Bicarbonate Ion is a polyatomic ion whose formula is HCO3-. Hydrogencarbonate is the carbon oxoanion resulting from the removal of a proton from carbonic acid.
How does carbonate affect pH?
Carbonate ion concentrations increase with increasing pH and when more CO2 dissolves in seawater it becomes more acidic. The bicarbonate and carbonate ions are responsible for the buffering capacity of seawater, i.e. seawater can resist drastic pH changes even after the addition of weak bases and acids.
What is the pH value of NaHCO3?
Baking soda, also known as sodium bicarbonate, has a pH of 9, making it a mildly alkaline substance.
How does bicarbonate affect pH?
If bicarbonate is reabsorbed and/or acid is secreted into the urine, the pH becomes more alkaline (increases). When bicarbonate is not reabsorbed or acid is not excreted into the urine, pH becomes more acidic (decreases).
Why does bicarbonate act as a base in water?
The statement you read that “baking soda is a base” comes from the fact that a solution of sodium bicarbonate (baking soda) and water has a pH of around 8.3. Again, in an otherwise pure aqueous system, it is the first reaction that dominates, and thus giving the observed slightly basic solution.
What is bicarbonate water?
Bicarbonate is a natural component of all mineral waters. Mineral waters that are sourced from limestone-rich areas typically have a high bicarbonate content. Bicarbonate plays a vital role in buffering acids and ensures that the mineral water tastes pleasantly clean and refreshing.
What is the pH of bicarbonate?
around 8.3
Baking soda, also known as sodium bicarbonate, is a base. This means that when people dissolve baking soda in water, it forms an alkaline solution. For example, a 0.1 molar solution of baking soda has a pH of around 8.3.
Is bicarbonate acidic or alkaline?
That box of baking soda, also known as bicarbonate of soda (or sodium bicarbonate), can do some major things. Its superpowers come from a two-letter term: pH. That stands for “power of hydrogen” to make something either an acid or a base (alkaline). Baking soda is an alkaline substance.
Why does carbonate increase pH?
Carbonate ion concentrations increase with increasing pH and when more CO2 dissolves in seawater it becomes more acidic. When CO2 from the atmosphere reacts with seawater, it immediately forms carbonic acid (H2CO3), which in itself is unstable. This further dissociates to form bicarbonate and carbonate ions.



