What happens during ATP hydrolysis?

Rachel Hickman
Published Feb 09, 2026
What happens during ATP hydrolysis?
ATP hydrolysis is the catabolic reaction process by which chemical energy that has been stored in the high-energy phosphoanhydride bonds in adenosine triphosphate (ATP) is released after splitting these bonds, for example in muscles, by producing work in the form of mechanical energy.
Does ATP hydrolyze in water?
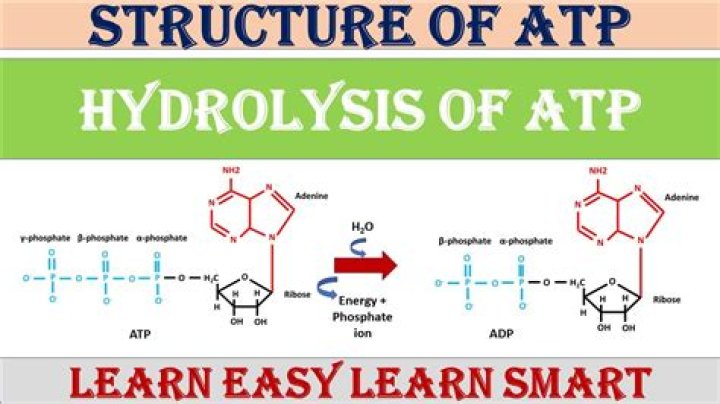
ATP can be hydrolyzed to ADP and Pi by the addition of water, releasing energy. ADP can be “recharged” to form ATP by the addition of energy, combining with Pi in a process that releases a molecule of water.
What is the function of ATP hydrolase?
Its main function is to store energy within the cell. This energy is released through an hydrolysis reaction, catalyzed by an enzyme called ATP hydrolase, in which ATP is decomposed in ADP (adenosin diphosphate) and Pi (phosphate). ATP hydrolysis is an exotermic reaction, releasing energy which is used by the cell.
What bonds does ATP have?
ATP is a nucleotide consisting of an adenine base attached to a ribose sugar, which is attached to three phosphate groups. These three phosphate groups are linked to one another by two high-energy bonds called phosphoanhydride bonds.
What is the role of ATP in energy coupling?
Essentially, the energy released from the ATP hydrolysis couples with the energy required to power the pump and transport Na+ and K+ ions. ATP performs cellular work using this basic form of energy coupling through phosphorylation. The sodium-potassium pump is an example of energy coupling.
What is the role of ATP in energy transfer?
The Adenosine triphosphate (ATP) molecule is the nucleotide known in biochemistry as the “molecular currency” of intracellular energy transfer; that is, ATP is able to store and transport chemical energy within cells. Energy is released by hydrolysis of the third phosphate group.
Does ATP hydrolysis release protons?
(The effect of these pK values is that a H+ is released on ATP hydrolysis, with a stoichiometry which approaches 1 above pK3′. This proton release can be used to assay the reactions of ATP hydrolysis or synthesis, or follow the kinetics if a recoding pH meter is available.)
How is the energy from ATP hydrolysis used?
The energy released from the hydrolysis of ATP into ADP is used to perform cellular work, usually by coupling the exergonic reaction of ATP hydrolysis with endergonic reactions.
How does ATP Resynthesised in cells?
ATP is resynthesised by the condensation of ADP and Pi. This reaction is catalysed by the enzyme ATP synthase during photosynthesis, or during respiration.
How does ATP serve as a carrier of free energy?
How does ATP serve as a carrier of free energy? It has unstable phosphate bonds that are easily broken; cells have enzymes that help break those bonds to release the energy. Regulated enzymes are allosteric; when a molecule binds to the allosteric site, the enzyme changes shape, which alters its activity.
How many bonds are in ATP?
ATP has two ‘high-energy’ bonds.
What bond stores the most energy in ATP?
Energy is stored in the covalent bonds between phosphates, with the greatest amount of energy (approximately 7 kcal/mole) in the bond between the second and third phosphate groups. This covalent bond is known as a pyrophosphate bond.