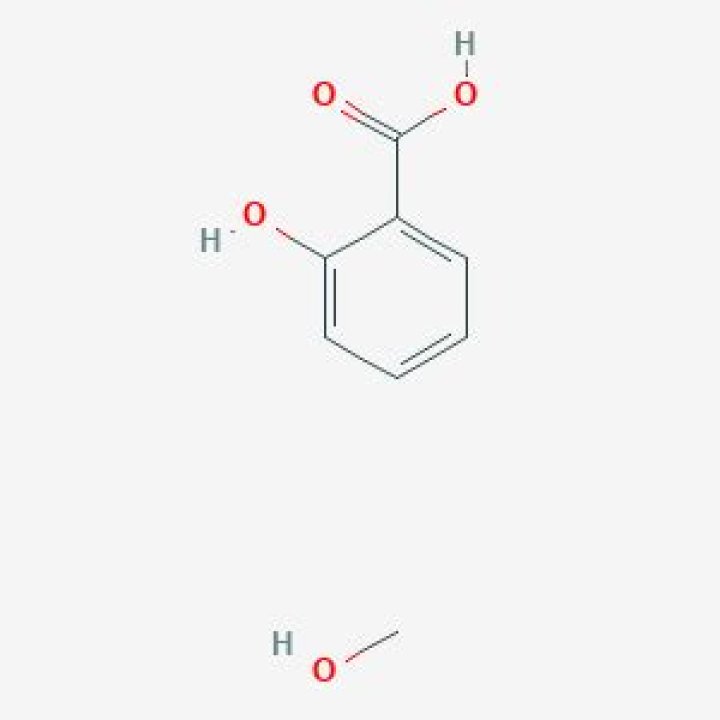
What does methanol and salicylic acid form

Emma Martin
Published Mar 31, 2026
When salicylic acid combines with methanol it becomes the ester known as methyl salicylate or oil of wintergreen. Methanol is also known as methyl alcohol and wood alcohol.
Does methanol react with salicylic acid?
The carboxylic group (-COOH) converts into ester (−COCH3) during the reaction of salicylic acid with methanol so, the reaction is known as esterification. The product methyl 2− hydroxyl benzoate is known as oil of wintergreen. Oil of wintergreen is a naturally produced organic ester.
What does ethanol and salicylic acid make?
Ethyl salicylate is the ester formed by the condensation of salicylic acid and ethanol. It is a clear liquid that is sparingly soluble in water, but soluble in alcohol and ether. It has a pleasant odor resembling wintergreen and is used in perfumery and artificial flavors.
What is the name of the ester formed from methanol and salicylic acid?
Methyl salicylate, an ester derived from the combination of salicylic acid and methanol, is also known as the oil of wintergreen.What does methanol and salicylic acid smell like?
It is the methyl ester of salicylic acid. It is a colorless, viscous liquid with a sweet, fruity odor reminiscent of root beer, but often associatively called “minty“, as it is an ingredient in mint candies. It is produced by many species of plants, particularly wintergreens.
Is methyl salicylate natural?
Methyl salicylate is a compound currently used in the creation of many flavors. It can be obtained by synthesis or from two natural sources: essential oil of wintergreen and essential oil of sweet birch bark.
What is methyl salicylate made from?
It derives from a salicylic acid. Methyl salicylate (oil of wintergreen or wintergreen oil) is an organic ester naturally produced by many species of plants, particularly wintergreens. The compound was first extracted and isolated from plant species Gaultheria procumbens in 1843.
What is methyl salicylate used for?
What is methyl salicylate topical? Methyl salicylate topical (for the skin) is used for temporary relief of muscle or joint pain caused by strains, sprains, arthritis, bruising, or backaches.How is methyl salicylate converted to salicylic acid?
Methyl salicylate (from oil of wintergreen,) and salicylic acid (from Latin salix, willow tree) are both natural products. Methyl salicylate can be converted into salicylic acid using the saponification reaction. You will perform this reaction and will purify the product using crystallization.
What functional group in salicylic acid reacts with methanol to form an ester?carboxylic acids In methyl salicylate (oil of wintergreen), the COOH group of salicylic acid is esterified with methanol (CH3OH), whereas in acetylsalicylic acid (aspirin) the acid component of the ester is acetic acid, and salicylic acid contributes the phenolic ―OH group.
Article first time published onWhat smell does methanol and Ethanoic acid make?
AlcoholOrganic acidSmell of esterPentanolEthanoic acidPearsOctanolEthanoic acidBananasPentanolButanoic acidStrawberriesMethanolButanoic acidPineapples
What is the product of methanol and butanoic acid?
The general word equation for the reaction is:alcohol (OH) + organic acid (COOH) → ester + waterFor example:methanol + butanoic acid → methyl butanoate + watermethyl= 1Cbutane= 4CThe alcohol (methanol) loose an H, and the carboxylic acid loose an OH which form the by product of this reaction = water The ester formed= …
What reacts with methanol and Methanoic acid?
Methanal is oxidised to methanoic acid by adding an oxygen atom. Methanol can also be oxidised by passing a mixture of methanol vapour and air over a silver catalyst at 500°C.
Is wintergreen oil the same as methyl salicylate?
Wintergreen oil is an essential oil that’s traditionally derived from the leaves of the wintergreen plant. Methyl salicylate, the active ingredient of wintergreen oil, can be chemically synthesized and is often referred to as wintergreen oil in many products.
Why is methyl salicylate toxic?
Methyl salicylate (oil of wintergreen) contains more salicylate than other salicylates; 5 ml methyl salicylate is equivalent to five aspirin tablets (325 mg each). The primary effects of salicylate toxicity are complex and include direct stimulation of the CNS respiratory center leading to a respiratory alkalosis.
Is salicylate the same as salicylic acid?
A salicylate is a salt or ester of salicylic acid. Salicylates are found naturally in some plants (such as white willow bark and wintergreen leaves) and are thought to protect the plant against insect damage and disease. Aspirin is a derivative of salicylic acid – and is also known as acetylsalicylic acid.
Is methyl salicylate the same as salicylic acid?
Methyl salicylate is a liquid methyl ester of salicylic acid with a distinct characteristic odor. … Therapeutic concentrations of methyl salicylate, averaged 4.9 mg/L at 15 rain after a 0.42-mL oral dose (4,5). Methyl salicylate is thought to be converted to salicylic acid, predominately by the liver.
Is methyl salicylate FDA approved?
Approval Date: 2/20/2008.
Is methyl salicylate the same as lidocaine?
Lidocaine is a local anesthetic (numbing medication). It works by blocking nerve signals in your body. Methyl salicylate is a nonsteroidal anti-inflammatory drug (NSAID) in a group of drugs called salicylates (sa-LIS-il-ates). This medicine works by reducing substances in the body that cause pain and inflammation.
Is methyl salicylate an aspirin?
Methyl salicylate (oil of wintergreen) is a chemical that smells like wintergreen. It is used in many over-the-counter products, including muscle ache creams. It is related to aspirin. Methyl salicylate overdose occurs when someone swallows a dangerous amount of a product containing this substance.
What is the side effects of methyl salicylate?
- redness or swelling of the treated area;
- increased pain; or.
- severe burning or skin irritation such as a rash, itching, pain, or blistering.
Where is methyl salicylate found?
Where is Methyl salicylate found? Methyl salicylate is used as a flavoring agent to provide fragrance to various products, and as an odor masking agent. It is found in household products, gum, candy, root beer, and mouthwashes.
Is salicylic acid an alcohol?
Note that the salicylic acid has an organic acid functional group, and an alcohol group, on an aromatic hydrocarbon ring. The acetylsalicylic acid retains the organic acid functionality, but the alcohol has been modified, to form an ester group.
How do you make salicylic acid?
Salicylic acid is produced commercially via the Kolbe-Schmitt process. Here phenol and sodium hydroxide are reacted to make sodium phenoxide. The phenoxide is contacted with CO2 to form sodium salicylate. The salicylate is acidified to give salicylic acid.
What drug class is salicylic acid?
Salicylic acid is in a class of medications called keratolytic agents. Topical salicylic acid treats acne by reducing swelling and redness and unplugging blocked skin pores to allow pimples to shrink.
How does topical methyl salicylate work?
Menthol and methyl salicylate are known as counterirritants. They work by causing the skin to feel cool and then warm. These feelings on the skin distract you from feeling the aches/pains deeper in your muscles, joints, and tendons.
Is salicylic acid a carcinogen?
Taken together, the data on these salicylates suggest that salicylic acid would have no carcinogenic potential.
Is methyl salicylate safe for babies?
Do not apply methyl salicylate topical to your breast area if you are breast-feeding a baby. Do not use this medication on a child younger than 12 years old without medical advice.
Why can salicylic acid form esters?
The major use of salicylic acid is in the preparation of its ester derivatives; since it contains both a hydroxyl (−OH) and a carboxyl (−CO2H) group, it can react with either an acid or an alcohol. … The hydroxyl group reacts with acetic acid to form the acetate ester, acetylsalicylic acid (see aspirin).
What is the purpose of Fischer esterification lab?
CourseOrganic Chemistry II Lab (CH 238) sulfuric acid, which is the catalyst, to develop an unknown ester. During the experiment, the Fischer Esterification purpose is to convert carboxylic acids to esters.
Is salicylic acid soluble in methanol?
… It has a very low solubility in water but is highly soluble in a wide range of organic solvents (methanol, ethanol, 2propanol and THF). Unlike its isomers (p-and m-hydroxybenzoic acid), no polymorphs or solvates of salicylic acid have been encountered [19] . …



