What does endothermic mean quizlet?

Christopher Anderson
Published Mar 04, 2026
What does endothermic mean quizlet?
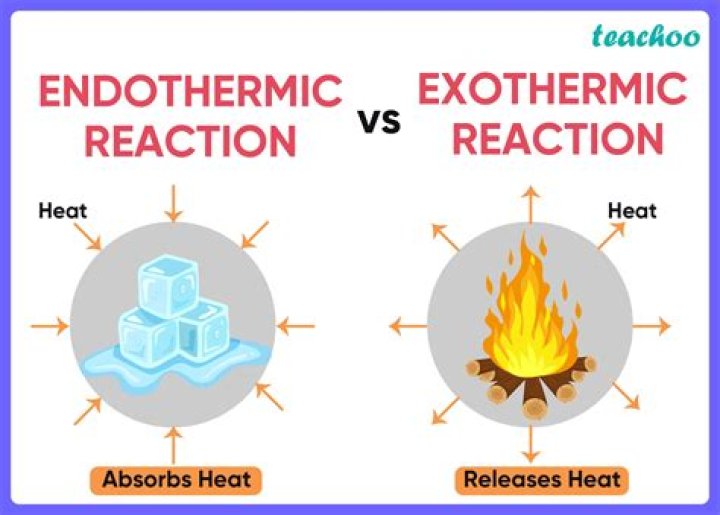
Endothermic reaction. A reaction in which energy is ABSORBED by matter. Energy flows into the system from the surroundings generally in the form of heat (heat is not being released, it’s being absorbed) (getting colder)
What is endothermic and exothermic quizlet?
A chemical reaction that releases energy, usually in the form of heat. An exothermic reaction releases energy and feels warm while an endothermic reaction absorbs energy and feels cool.
Which reactions are endothermic?
Endothermic Processes
- Melting ice cubes.
- Melting solid salts.
- Evaporating liquid water.
- Converting frost to water vapor (melting, boiling, and evaporation, in general, are endothermic processes.
- Making an anhydrous salt from a hydrate.
- Forming a cation from an atom in the gas phase.
- Splitting a gas molecule.
What is endothermic short answer?
Endothermic reactions are reactions that require external energy, usually in the form of heat, for the reaction to proceed. Therefore, the change in enthalpy is positive, and heat is absorbed from the surroundings by the reaction.
What occurs in endothermic reaction quizlet?
Endothermic reactions are reactions that absorb heat.
Which reaction is exothermic?
Chemical reactions that release energy are called exothermic. In exothermic reactions, more energy is released when the bonds are formed in the products than is used to break the bonds in the reactants. Exothermic reactions are accompanied by an increase in temperature of the reaction mixture.
What is the difference between exothermic and endothermic reactions?
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise. An endothermic process absorbs heat and cools the surroundings.”
Which is the neutralization reaction?
In chemistry, neutralization or neutralisation (see spelling differences) is a chemical reaction in which acid and a base react quantitatively with each other. In a reaction in water, neutralization results in there being no excess of hydrogen or hydroxide ions present in the solution.
What are 2 examples of exothermic reactions?
Here are some of the examples of exothermic reaction:
- Making of an ice cube. Making ice cube is a process of liquid changing its state to solid.
- Snow formation in clouds.
- Burning of a candle.
- Rusting of iron.
- Burning of sugar.
- Formation of ion pairs.
- Reaction of Strong acid and Water.
- Water and calcium chloride.
What does one mean by exothermic?
The chemical reactions release energy in the form of heat, light, or sound, are called exothermic reactions. An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat.
What is an endothermic reaction give one example?
Endothermic reaction: A chemical reaction in which heat is absorbed is called endothermic reaction. It causes fall in temperature. e.g. (i) When nitrogen and oxygen together are heated to a temperature of about 3000°C, nitric oxide gas is formed.
Which represents an endothermic reaction?
An endothermic reaction is a chemical reaction in which more energy is needed to break bonds in the reactants than is released when new bonds form in the products.
What is the difference between endothermic and exothermic reactions?
The difference between endothermic and exothermic reactions lies in the words themselves. “Thermic” refers to heat, just as in the word “thermometer.”. “Exo” means “outside” and “endo” means “inside.”. Thus, an endothermic reaction pulls heat into an object or area, while an exothermic reaction expels heat.
What is the reaction of K?
The k term is included in the rate law to denote the temperature dependency of the rate of reaction. The rate constant is the measure of reaction rate when the concentration of all the reactant are unity. It gives the measure of fastness of a reaction. It is denoted by k.
What are some examples of endothermic and exothermic reactions?
Examples of exothermic processes include burning of coal, rust formation and dissolution of quick lime in water. On the other hand, some endothermic processes include dissolution of Ammonium Chloride in water and nitric oxide formation. In chemical reactions, energy is required in the breaking up of atomic bonds.