What are zeolites in Engineering chemistry?

Emma Martin
Published Feb 15, 2026
What are zeolites in Engineering chemistry?
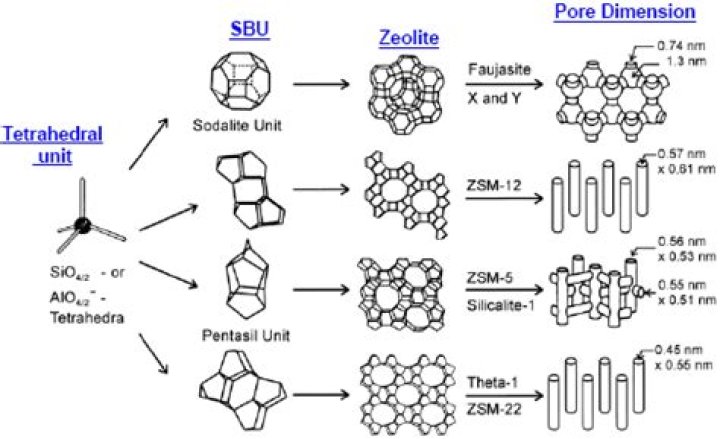
Zeolite structure Zeolites are a class of hydrated aluminum silicates made up of silica and alumina tetrahedra (SiO4 and AlO4 linked in a tetrahedral configuration). Zeolites’ uniform framework of pores can be exploited to control chemical reactions by adsorbing reactant molecules within the zeolite pores.
What is the structure of zeolite?
A zeolite is a hydrated aluminosilicate mineral with a structure characterized by a framework of linked tetrahedra, each consisting of four oxygen atoms surrounding a silicon or aluminum cation.
What is surfactant templating?
Surfactant-templating is one of the most effective and versatile synthetic strategies for the construction of well-defined porous architectures in solids. A thorough overview on the various strategies employing supramolecular chemistry to designing mesoporosity in zeolites is presented.
Why are zeolites used?
Uses of zeolites. Zeolites are widely used as ion-exchange beds in domestic and commercial water purification, softening, and other applications. In chemistry, zeolites are used to separate molecules (only molecules of certain sizes and shapes can pass through), and as traps for molecules so they can be analyzed.
What are zeolites and their types?
5.1. 1.3 Zeolite types
| Zeolite type | Pore size dimensions (Å) | Applications |
|---|---|---|
| Zeolite A | 4.1 | Detergent manufacturing |
| Faujasite | 7.4 | Catalytic cracking and hydrocracking |
| ZSM-5 | 5.2 × 5.8 | Xylene isomerization, benzene alkylation, catalytic cracking, catalyst dewaxing, and methanol conversion |
| Mordenite | 6.7 × 7.0 | Hydro-isomerization, dewaxing |
How do zeolites Work?
How does Zeolite work? Zeolite is an alkaline mineral that is very porous and has a negative charge. Since most toxins, such as heavy metals, radiation, and pesticides are positively charged, Zeolite is pulled towards toxins like a magnet and sucks them up into its structure.
What is a zeolite framework?
zeolite, any member of a family of hydrated aluminosilicate minerals that contain alkali and alkaline-earth metals. The essential structural feature of a zeolite is a three-dimensional tetrahedral framework in which each oxygen atom is shared by two tetrahedra.
What are the properties of zeolites?
Zeolites are aluminosilicate solids bearing a negatively charged honeycomb framework of micropores into which molecules may be adsorbed for environmental decontamination, and to catalyse chemical reactions. They are central to green-chemistry since the necessity for organic solvents is minimised.
What is soft templating?
The soft templating (also known as the endotemplate method) refers to supramolecular entities like self-assembled arrangements of structure-directing molecules like. Table 18.3. Mesoporosity Tailoring With Soft Templates. Pore Size (nm)
What is hard templating?
The hard templating (also known as the exotemplate method) implies the use of a solid material such as polymers, silica, and carbon [11]. First, mesoporous silica templates are tailored by surfactant self-assembly approaches, and then their pores are loaded with carbon sources such as sucrose.
What is the properties of zeolites?
Zeolites have two main properties: adsorption and ion exchange. These two properties are due to reactive surfaces, due to the presence of Al3+ on adsorption sites with a Si4+ ion resides, and the micropores crystalline system. These properties allow the zeolite for several applications.
How are zeolites classified?
Zeolites have been classified on the basis of their morphological characteristics, crystal structure, chemical composition, effective pore diameter, natural occurrence, etc.



