What are the 4 trends of the periodic table?

Robert Miller
Published Feb 13, 2026
What are the 4 trends of the periodic table?
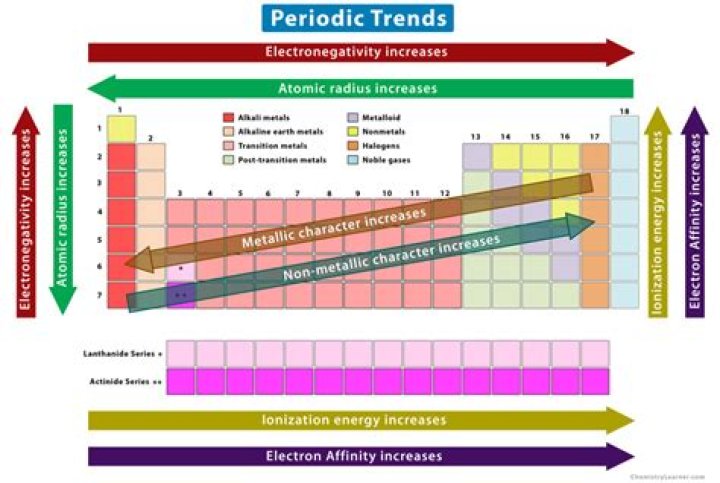
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character.
What are periodic trends in chemistry?
Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties.
How do you find the trend on the periodic table?
Explanation: Moving left to right on the periodic table causes an increase in atomic number (number of protons) as well as electron affinity and electronegativity. Atomic radius, however, will decrease when moving left to right. As more protons are added to the nucleus, they have a stronger attraction to the electrons.
How many trends are there in the periodic table?
The organization of the periodic table shows the periodic trends of six different physical properties of the elements: atomic radius, electron affinity, electronegativity, ionization energy, and metallic/nonmetallic character.
What are the trends in modern periodic table Class 10?
Trends In Modern Periodic Table
- Valency. 1.1 Variation of valency in a group. 1.2 Variation of valency in a period.
- Atomic Size. 2.1 Variation in a group. 2.2 Variation in a period.
- Metallic and Non-metallic Properties. 3.1 Metallic Character. 3.2 Non-metallic character.
- Electronegativity.
What trends are found in the groups families of the periodic table?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties.
How does periodic trends affect the elements?
Periodic trends affect bonding, because of how the elements are arranged on the periodic table. For example elements can be arranged by their electronegative, electron affinity, atomic radius, or ionization energy. Electronegative is the atoms ability to attract other bonded atoms.
How do trends in the periodic table help predict the properties of an element?
The Periodic Table can predict the properties of new elements, because it organizes the elements according to their atomic numbers. Creating new elements is not a simple process. When these heavy elements form, they are usually highly unstable. They decay so quickly that we don’t usually see the element itself.
What is the trend as we go down group 1?
Going down the group, the first ionisation energy decreases. There is more shielding between the nucleus and the outer electrons and the distance between the nucleus and the outer electron increases and therefore the force of attraction between the nucleus and outer most electrons is reduced.
What is periodic trends in properties of elements Class 11?
Periodic trends are those which explain certain properties of elements that are present in the periodic table. The periodic trends are electronegativity, ionic radius, atomic radius, electron affinity, ionization energy, chemical reactions of elements, and metallic character.
What are the trends of group 8 elements?
Noble Gases: Trends
- The noble gases have weak van Der Waals forces within their atoms.
- As non-metals, the noble gases have low boiling points.
- Atomic mass increases moving down the column.
- The density of the noble gases also changes moving down the column.
What are the trends in group 1?
Explaining the trend The reactivity of group 1 elements increases as you go down the group because: the atoms become larger. the outer electron becomes further from the nucleus. the force of attraction between the nucleus and the outer electron decreases.
What are the trends across the periodic table?
Major periodic trends include:electronegativity, ionization energy, electron affinity, atomic radius, melting point, andmetallic character. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element’s properties.
What does the periodic table have to do with chemistry?
The periodic-table. noun. The periodic table is a chart used in chemistry that shows chemical elements in order of their atomic number. An organized table of chemical elements such as oxygen and carbon is an example of the periodic table.
How do chemists use the periodic table?
It’s about seeing trends and patterns in the ways elements behave — that’s why it’s the periodic table after all. One of the most common ways chemists use it is to identify chemical similarities among elements, typically by considering elements in the same column of the periodic table.
What are solubility trends in the periodic table?
5.7: Predicting Solubility Trends Salts of the alkali metal ions and the ammonium ion, Li +, Na +, K +, and NH 4 + are almost always soluble. Virtually all metal nitrates and metal acetates are soluble. Metal halides are generally soluble, except for salts of Ag +, Pb 2+, Cu + and Hg +. Metal sulfates are generally soluble, except for salts of Ba 2+, Pb 2+ and Ca 2+.