What are physical and chemical properties of neon?

John Castro
Published Feb 13, 2026
What are physical and chemical properties of neon?
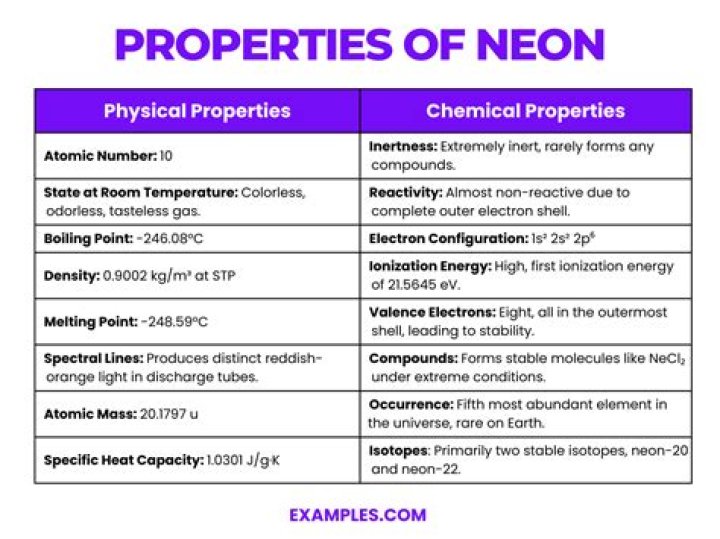
Neon appears as a colorless odorless noncombustible gas. Chemically inert.
What are the properties and uses of neon?
Neon is also used to make high-voltage indicators and switching gear, lightning arresters, diving equipment and lasers. Liquid neon is an important cryogenic refrigerant. It has over 40 times more refrigerating capacity per unit volume than liquid helium, and more than 3 times that of liquid hydrogen.
What are the physical and chemical properties of each element?
Physical properties include color, density, hardness, and melting and boiling points. A chemical property describes the ability of a substance to undergo a specific chemical change. To identify a chemical property, we look for a chemical change.
Which element has the same chemical properties as neon?
Explanation: Helium, neon, and xenon are in the same group, which are the noble gases (group 18/VIIIA). All of the atoms of the noble gases have filled valence shells–two valence electrons for helium, and eight valence electrons for the rest. This makes them very non-reactive, which is a hallmark of the noble gases.
Is neon chemically reactive?
Neon, along with helium, argon, krypton and xenon, make up the group known as noble gases. These are the most stable and least reactive elements due to having full valence shells (the outer shell has the max number of electrons, two for helium, eight for the rest).
Which is chemical property?
A chemical property is a characteristic of a particular substance that can be observed in a chemical reaction. Some major chemical properties include flammability, toxicity, heat of combustion, pH value, rate of radioactive decay, and chemical stability.
What is neon chemical properties?
The key properties of neon include the following: It is a colorless, tasteless odorless inert gas. It changes to reddish-orange color in vacuum tube. It is chemically inactive. It has the lowest liquid range of any element.
What are some chemical properties of an element?
What are physical and chemical properties of water?
Hydrolysis reaction
| Properties | |
|---|---|
| Odour | None |
| Density | Solid: 0.9167 g/ml at 0 °C Liquid: 0.961893 g/mL at 95 °C 0.9970474 g/mL at 25 °C 0.9998396 g/mL at 0 °C |
| Boiling point | 99.98 °C (211.96 °F; 373.13 K) |
| Melting point | 0.00 °C (32.00 °F; 273.15 K) |
What are some chemical properties of neon?
The key properties of neon include the following:
- It is a colorless, tasteless odorless inert gas.
- It changes to reddish-orange color in vacuum tube.
- It is chemically inactive.
- It has the lowest liquid range of any element.
What is chemically similar to neon?
Noble gas
NonmetalPeriod 2 element
Neon/Chemical series
These gases all have similar properties under standard conditions: they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six noble gases that occur naturally are helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn).
What are chemical properties of neon?
What are some general properties of neon?
Color : Colorless – under low pressure,it glows a bright orange-red if an electric current is passed through it
What are the chemical properties of the element neon?
Neon is a chemical element with symbol Ne and atomic number 10. It is chemically inert and forms no uncharged chemical compounds. The only true property that can be associated with this rare gas is that it glows red when charged with high voltage electricity in a vacuum tube.
What are the characteristics of neon?
Symbol: Ne
What are 3 examples of chemical properties?
Examples of chemical properties. Reactivity against other chemical substances. Heat of combustion. Enthalpy of formation. Toxicity. Chemical stability in a given environment. Flammability. Preferred oxidation state(s)