Is water a reactant or product in dehydration synthesis?

Christopher Anderson
Published Mar 16, 2026
Is water a reactant or product in dehydration synthesis?
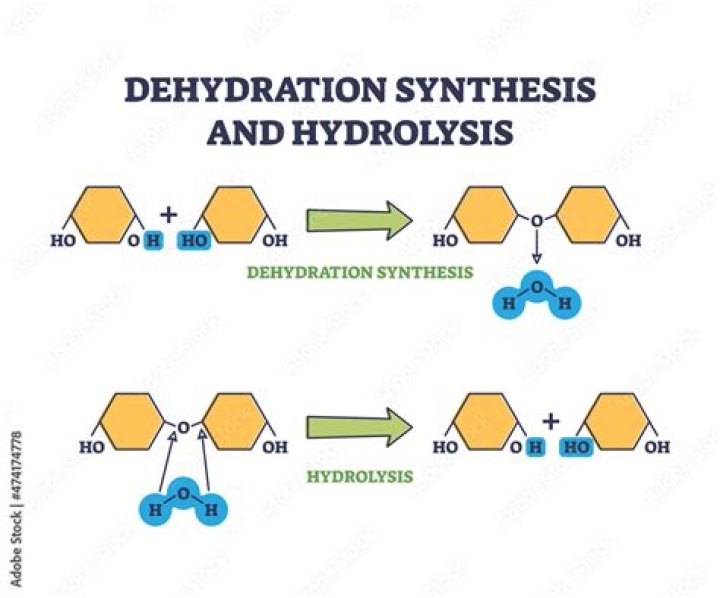
The Chemistry of Dehydration Synthesis 1. Two reactants combine to form a new product. 2. Water is lost as a result of the reaction.
Is water part of a dehydration reaction?
In chemistry, a dehydration reaction, also known as Zimmer’s Hydrogenesis, is a chemical reaction that involves the loss of water from the reacting molecule or ion. It is the most common type of condensation reaction. Dehydration reactions are common processes, the reverse of a hydration reaction.
Does dehydration produce water as a by product?
Dehydration Synthesis The monomers combine with each other via covalent bonds to form larger molecules known as polymers. In doing so, monomers release water molecules as byproducts.
What is the by product of a dehydration reaction?
In doing so, monomers release water molecules as byproducts. In a dehydration synthesis reaction between two un-ionized monomers, such as monosaccharide sugars, the hydrogen of one monomer combines with the hydroxyl group of another monomer, releasing a molecule of water in the process.
Is dehydration exothermic or endothermic?
Typically, all Dehydration Synthesis reactions, where smaller compounds are bonded together to produce larger compounds, are endothermic.
What is water dehydration?
Overview. Dehydration occurs when you use or lose more fluid than you take in, and your body doesn’t have enough water and other fluids to carry out its normal functions. If you don’t replace lost fluids, you will get dehydrated.
What produces water as a by product?
Water is produced by our bodies as a byproduct of the metabolism during a process called cellular respiration, in which glucose and oxygen are used to produce energy for cells.
What is the role of water in a dehydration reaction?
Dehydration reactions link monomers together into polymers by releasing water, and hydrolysis breaks polymers into monomers using a water molecule. Monomers are just single unit molecules and polymers are chains of monomers.
Are dehydration reaction exothermic?
The rate of dehydration then accelerates as the acid heats up because the reaction is exothermic. As the sugar molecules are stripped of water, the heat generated turns the water into steam which then expands the remaining carbon into a porous, smoking, black column.
Is dehydrating an agent?
A dehydrating agent is a substance that dries or removes water from a material. Sulfuric acid, concentrated phosphoric acid, hot aluminum oxide, and hot ceramic are common dehydrating agents in these types of chemical reactions.
What happens dehydration?
Dehydration occurs when your body loses more fluid than you take in. When the normal water content of your body is reduced, it upsets the balance of minerals (salts and sugar) in your body, which affects the way it functions. Water makes up over two-thirds of the healthy human body.
Can dehydration cause pins and needles?
Nausea or feeling sick. Constipation. Tingling or numbness in fingers or toes or a feel of body parts “falling asleep” Lack of – or reduced – sweating, even in strenuous situations.
What is the definition of dehydration reaction?
Dehydration Reaction Definition. A dehydration reaction is a chemical reaction between two compounds where one of the products is water. For example, two monomers may react where a hydrogen (H) from one monomer binds to a hydroxyl group (OH) from the other monomer to form a dimer and a water molecule (H2O).
Which of the following is a dehydrating agent?
The reverse reaction, where water combines with hydroxyl groups, is termed hydrolysis or a hydration reaction. Chemicals commonly used as dehydrating agents include concentrated phosphoric acid, concentrated sulfuric acid, hot ceramic and hot aluminum oxide. Also Known As: A dehydration reaction is the same as a dehydration synthesis.
What happens in the dehydration synthesis reaction between two amino acids?
In the dehydration synthesis reaction between two amino acids, with are ionized in aqueous environments like the cell, an oxygen from the first amino acid is combined with two hydrogens from the second amino acid, creating a covalent bond that links the two monomers together to form a dipeptide. In the process a water molecule is formed.
What is the relative reactivity of alcohols in dehydration reactions?
The relative reactivity of alcohols in dehydration reactions is ranked as follows: Primary alcohols dehydrate through the E2 mechanism. The hydroxyl oxygen donates two electrons to a proton from sulfuric acid (H 2 SO 4 ), forming an alkyloxonium ion.