Is TLC polar

John Castro
Published Apr 02, 2026
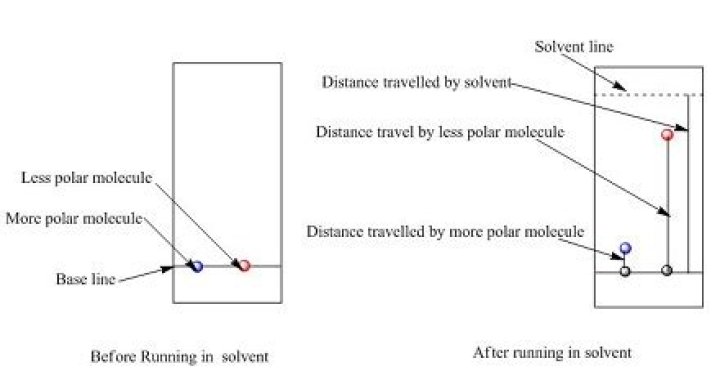
The stronger a compound is bound to the adsorbent , the slower it moves up the TLC plate. Non-polar compounds move up the plate most rapidly (higher Rf value), whereas polar substances travel up the TLC plate slowly or not at all (lower Rf value).
Is TLC polar or nonpolar?
This very polar stationary phase is paired with a relatively nonpolar mobile phase (an organic solvent or solution), in what is referred to as “normal phase” TLC.
Does TLC have a polar stationary phase?
TLC most often uses polar silica gel, a form of silicon dioxide, as the stationary phase. The stationary phase forms hydrogen bonds due to the OH groups on its surface.
How does polarity relate to TLC?
The more polar the compound, the more it will adhere to the adsorbent and the smaller the distance it will travel from the baseline, and the lower its Rf value.Which is more polar on TLC?
Answer: Alumina is more polar than is silica (see the first paragraph under “The Adsorbent” in the TLC section). Therefore, each of the compounds would travel slower on an alumina TLC plate than on a silica TLC plate, and the Rf values for each of the compounds would be smaller.
Is benzaldehyde a polar molecule?
Benzaldehyde has an aromatic ring (with three π bonds), which is non-polar.
What is TLC absorbent?
Thin layer chromatography (TLC) is an affinity-based method used to separate compounds in a mixture. … In TLC, the stationary phase is a thin adsorbent material layer, usually silica gel or aluminum oxide, coated onto an inert plate surface, typically glass, plastic, or aluminum.
How do you know if a pigment is polar?
More polar pigments that have residual charges (like water) will not interact much with the solvent, staying closer to the bottom of the strip. High Rf values from TLC using a nonpolar solvent means the pigment is more nonpolar. Lower Rf values mean the pigment is more polar.How polar is chlorophyll?
Both chlorophylls contain C—O and C—N bonds (polar groups) and also contain magnesium bonded to nitrogen – forming a bond so polar that it is almost ionic. The structures of the chlorophylls are given below. Both chlorophylls are much more polar than β-carotene.
Why must the TLC plate be handled on the edges?Aluminum and plastic plates are also flexible, which may result in flaking of the stationary phase. Never under any circumstances touch the face of a TLC plate with your fingers as contamination from skin oils or residues on gloves can obscure results. Instead, always handle them by the edges, or with forceps.
Article first time published onWhat is TLC protocol?
Thin layer chromatography (TLC) is a quick, sensitive, and inexpensive technique used to determine the number of components in a mixture, verify the identity and purity of a compound, monitor the progress of a reaction, determine the solvent composition for preparative separations, and analyze the fractions obtained …
Is TLC partition chromatography?
We very often use thin layer chromatography in laboratories. The spots are made visible by UV light. Complete step by step answer: … Partition chromatography is the method to separate the components between two liquid phases using a column.
What is the principle of TLC?
The principle of TLC is the distribution of a compound between a solid fixed phase (the thin layer) applied to a glass or plastic plate and a liquid mobile phase (eluting solvent) that is moving over the solid phase.
What is TLC plate made of?
TLC also allows for some qualitative conclusions to be drawn regarding the nature (polarity) of a compound by calculating that component’s Rf value. The thin-layer chromatography plate is composed of silica gel (the adsorbent ) adhered to a plastic backing for support. Reminder: silica is a polar compound.
Is acetaminophen a polar?
Acetaminophen has a phenol and amide function group, but caffeine has multiple amide functional groups; therefore, acetaminophen is more polar than aspirin and less polar than caffeine.
Is aspirin a polar?
Aspirin is a polar molecule with dipole-dipole attraction bonds and an -OH (hydroxyl) segment as part of a carboxylic acid group.
Is TLC quantitative or qualitative?
Thin-layer chromatography (TLC) is a widely used method for qualitative analysis to determine the number of components in a mixture, to determine the identity of two substances, or to monitor the progress of a reaction. The more accurate high-performance TLC (HPTLC) is better suited for quantitative analysis.
What is the solid support in TLC?
TLC is composed of two phases, a mobile and a solid phase. The solid phase is a thin solid support that usually consists of Alumina or Silica. The mobile phase is a solvent that moves through capillary action right through the solid phase.
How does TLC work to separate compounds?
Thin layer chromatography, or TLC, is a method for analyzing mixtures by separating the compounds in the mixture. … Development consists of placing the bottom of the TLC plate into a shallow pool of a development solvent, which then travels up the plate by capillary action.
Why is benzaldehyde not soluble in water?
The lone pair on oxygen atom of benzaldehyde is completely involved in conjugation making it less available to form hydrogen bonding with water.
How do you determine polarity?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
Which pigment is more polar?
PigmentRf valueChlorophyll a0.59Chlorophyll b0.42Anthocyanins0.32-0.62Xanthophylls0.15-0.35
Is chlorophyll soluble in water?
Chlorophyll is the pigment that gives plants and algae their green color. … The long hydrocarbon (phytol) tail attached to the porphyrin ring makes chlorophyll fat-soluble and insoluble in water.
Are anthocyanins polar?
The pigments represent a wide range of polarity from the chlorophylls and carotenoids which are very water insoluble (hydrophobic or nonpolar) and found embedded in membranes to the anthocyanins which are very water soluble (hydrophilic or polar).
What pigment is most non-polar?
Carotene moves the farthest because it is the most nonpolar of the pigments and it is attracted more strongly to the acetone-ligroin mixture (mobile phase) than to the paper. This stronger, nonbonded interaction with the mobile phase indicates that carotene is the most nonpolar pigment found in spinach chloroplasts.
Is yellow pigment polar?
The yellow-green pigment is chlorophyll β. The orange-yellow pigment is carotene and the light yellow pigments are called xanthophylls. These four pigments are non-polar and are not soluble in water.
Is blue pigment polar?
Blue is the least polar pigment.
What happens if TLC plate is slanted?
If the plate enters the solvent at an angle, it will cause a slanted solvent front line which may result in poor separation and uneven alignment of the spots.
Do polar compounds move slower up TLC plate?
The stronger a compound is bound to the adsorbent , the slower it moves up the TLC plate. Non-polar compounds move up the plate most rapidly (higher Rf value), whereas polar substances travel up the TLC plate slowly or not at all (lower Rf value).
What will be the appearance of a TLC plate if a solvent of too low polarity is used for the development a solvent of too high polarity?
If a development solvent of too high a polarity is used, all components in the mixture will move along with the solvent and no separation will be observed (Rf’s will be too large). If the solvent is of too low a polarity the components will not move enough, and again separation will not occur (Rf’s will be too small).
What type of chromatography is TLC?
TechniqueStationary phase*Thin layer chromatography (TLC)solid (silica or alumina)*Liquid column chromatographysolid (silica or alumina)Size exclusion chromatographysolid (microporous beads of silica)Ion-exchange chromatographysolid (cationic or anionic resin)