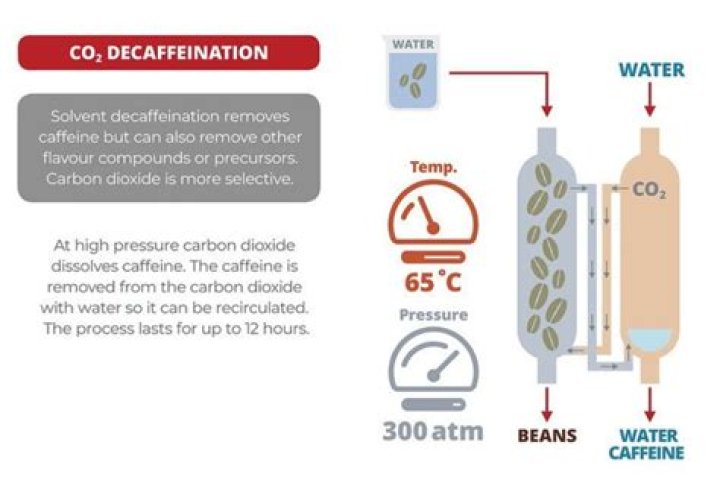
Is supercritical carbon dioxide decaffeination safe?

Daniel Johnson
Published Feb 11, 2026
Is supercritical carbon dioxide decaffeination safe?
Supercritical carbon dioxide is especially well-suited to the decaffeinating process, since it removes caffeine while leaving the aroma oils, and the beans do not need to be treated after decaffeination to remove solvent residues. The carbon dioxide can be stripped of caffeine residue and reused.
Why is CO2 used in supercritical fluids?
Carbon dioxide (CO2) is the most widely used supercritical fluid. This is because CO2 is cheap, chemically inert, non-toxic, non-flammable and readily available at high purities and at low costs.
What conditions must be used to form supercritical carbon dioxide?
CO2 exists at supercritical condition above a critical temperature (Tc = 304.1 K) and pressure (Pc = 73.8 bar). It has the properties of both a gas and a liquid in this state. By changing the temperature and pressure, the phase changes from solid to liquid and then to gas.
Why is supercritical CO2 a good solvent?
Supercritical CO2 is a unique solvent that has the characteristics of variable density, low viscosity, and high diffusivity. The manipulation of these characteristics has led to numerous applications of this green solvent in diverse areas including extractions, impregnations, particle formation, and cleaning.
How do you Decaffeinate coffee beans?
The most-common methods of decaffeination involve chemical solvents, usually ethyl acetate or methylene chloride. In the direct method, the coffee beans are steamed and then rinsed repeatedly with the chemical solvent to flush away the caffeine.
How do you Decaffeinate yourself?
What you can do to feel better
- No more caffeine. Don’t consume any more caffeine today.
- Drink plenty of water. Caffeine is a diuretic, which means that you need to drink extra water to make up for what you’re peeing out.
- Replace electrolytes.
- Take a walk.
- Practice deep breathing.
What is subcritical CO2?
Subcritical (low temperature, low pressure) CO2 extractions take more time and produce much smaller yields than supercritical, but they keep essential oils, terpenes and other sensitive chemicals in the plant.
What is supercritical CO2 used for?
Supercritical carbon dioxide is used to remove organochloride pesticides and metals from agricultural crops without adulterating the desired constituents from the plant matter in the herbal supplement industry.
What happens supercritical point?
A supercritical fluid (SCF) is any substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist, but below the pressure required to compress it into a solid. …
What is natural decaffeination?
The challenge in decaffeination is removing the caffeine chemical without diminishing, altering, or removing other naturally occurring chemicals that create ‘coffee’. …
How do they Decaffeinate tea?
The most common decaffeination method involves soaking tea leaves in a chemical solvent–either ethyl acetate or methylene chloride. Teas decaffeinated with ethyl acetate are often labeled “naturally decaffeinated” because this chemical compound is derived from fruit.