Is strontium chromate soluble in water

Daniel Johnson
Published Apr 21, 2026
Strontium chromate is a light yellow powder or granular solid. It is insoluble in water. The primary hazard is the threat to the environment.
What is the solubility of strontium chromate?
NamesSolubility in water0.12 g/100 mL (15 °C) 3 g/100 mL (100 °C)SolubilitySoluble in dilute acids and ammoniaMagnetic susceptibility (χ)−5.1·10−6 cm3/molStructure
Is na2cro4 aqueous?
Sodium chromate is a yellow crystalline solid dissolved in a liquid medium, probably water. It is soluble in water. It is toxic by inhalation, ingestion and/or skin contact.
Are strontium salts soluble in water?
Strontium is water insoluble, but it does react with water. Strontium compounds can be water soluble. Examples include strontium carbonate with a water solubility of 10 mg/L, and strontium chromate with a water solubility of 9 mg/L.Is magnesium chromate soluble or insoluble?
Magnesium chromate is a chemical compound, with the formula MgCrO4. It is a yellow, odorless, water-soluble solid with several important industrial uses. This chromate can be manufactured as a powder.
Is strontium chromate the same as hexavalent chromium?
Hexavalent chromium (CrVI) is present in the conversion coat in the form of chromic acid and in the primer in the form of strontium chromate. … In the absence of a change from chromated to nonchromated conversion coats and primers, additional control measures are needed to reduce these exposures.
Is bacro4 soluble in water?
It is very insoluble in water, but is soluble in acids: 2 BaCrO4 + 2 H+ → 2 Ba2+ + Cr2O72− + H2O. Ksp = [Ba2+][CrO42−] = 2.1 × 10. It can react with barium hydroxide in the presence of sodium azide to create barium chromate(V).
Is strontium chloride soluble in water?
Strontium Chloride is an excellent water soluble crystalline Strontium source for uses compatible with chlorides. Chloride compounds can conduct electricity when fused or dissolved in water.Is strontium hydroxide soluble?
According to handbook data (Merck index and CRC handbook) strontium hydroxide is very soluble in water (>10g/L) at 25°C.
Is strontium sulphate soluble in water?Strontium sulfate (SrSO4) is the sulfate salt of strontium. It is a white crystalline powder and occurs in nature as the mineral celestine. It is poorly soluble in water to the extent of 1 part in 8,800.
Article first time published onIs nacro4 soluble?
Chromates (CrO4 2-) are usually insoluble. Exceptions include Na2CrO4, K2CrO4, (NH4)2CrO4, and MgCrO4. … Exceptions include salts of the Na+, K+, and NH4 + ions, which are soluble by rule 1.
Is Ammonium chromate soluble in water?
A yellow crystalline solid. Density 1.866 g / cm3. Soluble in water. Toxic by inhalation (of dust).
Is copper chromate soluble in water?
copper chromate Chemical Properties,Uses,Production The neutral form is a reddish-brown crystalline solid; decomposes slowly to copper(II) chromite when heated above 400°C; insoluble in water; soluble in acids.
Is bahco3 soluble?
Barium carbonate is the inorganic compound with the formula BaCO3. Like most alkaline earth metal carbonates, it is a white salt that is poorly soluble in water.
Is calcium chromate soluble in water?
Calcium chromate is a yellow powder. It is slightly soluble in water.
Is Lithium chloride soluble?
CompoundWater Solubility in g/100 mL at 20oCLiCl83LiBr166LiI150
Is bacro4 more soluble than BaCr2O7?
Why? Because the dichromate is larger than the chromate, and thus LESS charge dense, and should thus be LESS effectively solvated in an aqueous medium, and the ionic bond between cation and dianion should be weaker. Entropy and enthalpy would suggest that BaCr2O7 is the more soluble.
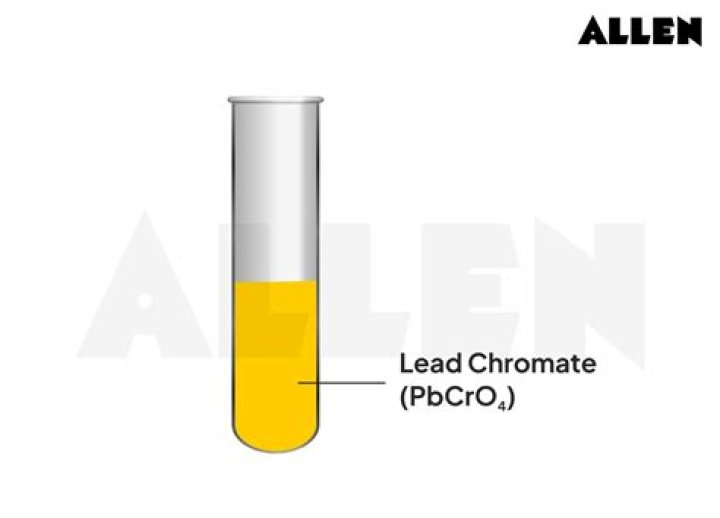
Is pbcro4 soluble or insoluble?
NamesSolubility in waternegligibleSolubilitysoluble in diluted nitric acid insoluble in acetic acid, ammoniaMagnetic susceptibility (χ)−-18.0·10−6 cm3/molRefractive index (nD)2.31
Is pbl2 soluble in water?
Lead iodide appears as a yellow crystalline solid. Insoluble in water and denser than water.
What is strontium chromate used for?
Strontium Chromate is a light yellow powder. It is used as a metal protective coating to prevent corrosion, as a colorant in polyvinyl chloride resins, and in pyrotechnics.
Why is chromium used in aircraft?
Generally referred to as chrome, chromate, or chromium, hexavalent chromium is found in the surface preparation processes and primers of traditional aircraft paint systems. Chromates function as “sacrificial anodes,” or metals that protect an aircraft’s aluminum skin by reacting to corrosion first.
Is strontium hydroxide more soluble in water than calcium hydroxide?
ElementLattice Energy (kJ/mol)Coordination NumberSr24748Ba23308
Is strontium nitrate soluble in water?
Strontium Nitrate is a highly water soluble crystalline Strontium source for uses compatible with nitrates and lower (acidic) pH.
Why is strontium hydroxide less soluble in hot water?
Why does this happen at all? Its somewhat complex but the simple version of it is that these hardness ions reject heat when they dissolve in a solution. The hotter a fluid gets, the harder it is to reject that heat into the solution which forces the equilibrium back towards the solid form.
Why does strontium chloride dissolve in water?
NamesSolubility in wateranhydrous: 53.8 g/100 mL (20 °C) hexahydrate: 106 g/100 mL (0 °C) 206 g/100 mL (40 °C)Solubilityethanol: very slightly soluble acetone: very slightly soluble ammonia: insolubleMagnetic susceptibility (χ)−63.0·10−6 cm3/mol
What type of bond does strontium chloride have?
If the electronegativity difference is greater than 1.7 it has greater than 50% ionic character and is usually considered an ionic bond.
Is strontium chloride hygroscopic?
Strontium chloride is a white hygroscopic cubic crystalline or a colourless cubic crystalline solid substance and strontium chloride hexahydrate is a colourless, odourless crystals or white granule.
Is srcro4 soluble?
Strontium chromate is a light yellow powder or granular solid. It is insoluble in water.
Is strontium sulfate soluble in acid?
Strontium sulfate Properties aqueous acid: slightly soluble(lit.) Soluble in dilute hydrochloric acid and nitric acid . Slightly soluble in water < 0.01 g/L . Insoluble in ethanol and alkalis.
Does CaS dissolve in water?
The sulfides of all metals except barium, calcium, magnesium, sodium, potassium, and ammonium are insoluble in water. BaS, CaS, and MgS are sparingly soluble. … These basic salts are soluble in dilute acids but are not soluble in water.
Is alpo4 soluble?
Aluminum phosphate, solution appears as a colorless liquid. Insoluble in water.



