Is N2 a homonuclear molecule?

Sarah Cherry
Published Mar 04, 2026
Is N2 a homonuclear molecule?
Homonuclear diatomic molecules include hydrogen (H2), oxygen (O2), nitrogen (N2) and all of the halogens.
What are the period 2 homonuclear diatomic molecules?
Homonuclear diatomic molecules in Period 2: N2, O2, F.
What are the 8 homonuclear diatomic molecules?
The elements found as diatomic molecules are hydrogen (H, element 1), nitrogen (N, element 7), oxygen (O, element 8), fluorine (F, element 9), chlorine (Cl, element 17), bromine (Br, element 35), and iodine (I, element 53).
Why do homonuclear diatomic molecules always form nonpolar bonds?
Diatomic molecules are composed of only two atoms, of either the same or different chemical elements. The bond in a homonuclear diatomic molecule is non-polar due to the electronegativity difference of zero.
What is homonuclear and heteronuclear molecule?
These diatomic molecules can be classified as either homonuclear, meaning that they contain two atoms of the same element, or heteronuclear, which requires that they be comprised of one atom of two different elements.
What compound is N2?
Nitrogen | N2 – PubChem.
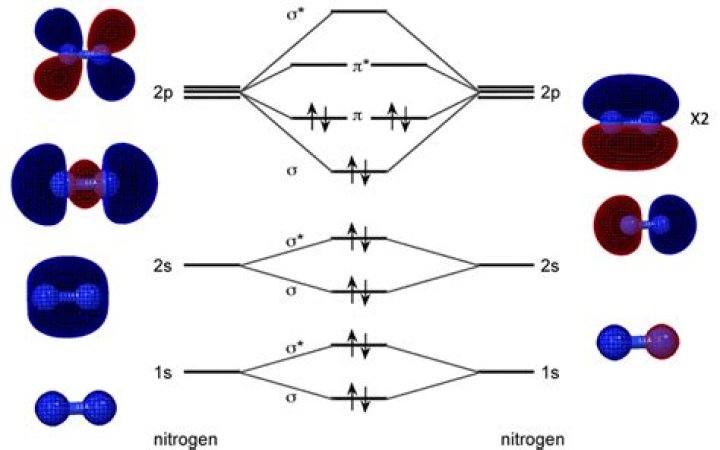
How many molecular orbitals are in N2?
N2 molecule had 14 electrons and 7 OCCUPIED Orbitals.
What is homonuclear and heteronuclear?
Which homonuclear molecules of the second period should have zero bond order?
The homo nuclear molecules of Lithium and Berilium in the second period will have zero bond order. Explanation: Homonulcear molecules are those molecules which are composed on only one type of element.
What is the difference between a homonuclear and heteronuclear molecule?
The key difference between homonuclear and heteronuclear diatomic molecules is that homonuclear diatomic molecules contain two atoms of the same element whereas heteronuclear diatomic molecules contain two atoms of different elements.
Which is not the homonuclear molecule?
Which of the following molecule is not homonuclear? Explanation: NO is Heteronuclear diatomic molecule in which atomic number of nitrogen is 7 and that of oxygen is 8 i.e., total number of electrons = 15.



