Is D fructose a furanose?

Daniel Johnson
Published Feb 11, 2026
Is D fructose a furanose?
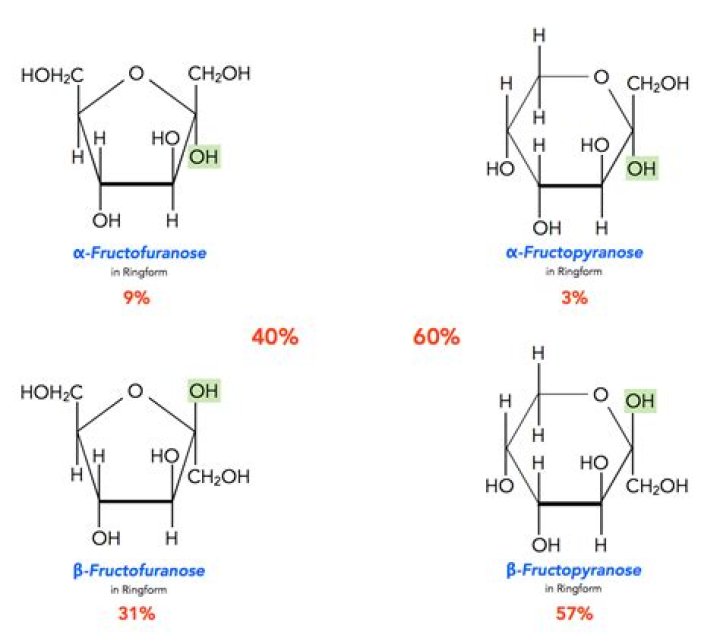
Monosaccharides in solution exist as equilibrium mixtures of the straight and cyclic forms. In solution, glucose is mostly in the pyranose form, fructose is 67% pyranose and 33% furanose, and ribose is 75% furanose and 25% pyranose.
What is beta D fructose?
Description. Beta-D-fructofuranose is a D-fructofuranose. It has a role as a mouse metabolite. It is an enantiomer of a beta-L-fructofuranose.
Why fructose is called furanose?
A furanose is a collective term for carbohydrates that have a chemical structure that includes a five-membered ring system consisting of four carbon atoms and one oxygen atom. The name derives from its similarity to the oxygen heterocycle furan, but the furanose ring does not have double bonds.
How does D glucose form pyranose and furanose?
Formation. The pyranose ring is formed by the reaction of the hydroxyl group on carbon 5 (C-5) of a sugar with the aldehyde at carbon 1. This forms an intramolecular hemiacetal. If reaction is between the C-4 hydroxyl and the aldehyde, a furanose is formed instead.
What are the examples of furanose?
| Classification | Example |
|---|---|
| Furanose | Fructose |
| Pyranose | b-D-glucose |
What is the Iupac name of fructose?
Fructose
Fructose/IUPAC ID
What is the structure of alpha D fructose?
D-Fructose
| PubChem CID | 2723872 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | C6H12O6 |
| Synonyms | D-Arabino-Hex-2-ulo-Pyranose D-Fru D-Fructopyranose D-Fructopyranoside D-Fructose More… |
| Molecular Weight | 180.16 |
What is furanose and pyranose?
Furanoses and Pyranoses Cyclic sugars that contain a five membered ring are called “furanoses”. The term is derived from the similarity with the aromatic compound furan and tetrahydrofuran. Cyclic sugars that contain a six membered ring are called “pyranoses”
Why is D fructose Levorotatory?
When fructose solution is placed in a polariser it rotates the plane polarized light in anti-clockwise direction which is towards left side and thus, it is a laevorotatory compound.
Can glucose form a furanose?
Glucose Has Several Structures, All In Equilibrium With Each Other. We’ve seen five separate isomers so far: the straight chain form, the pyranose form (alpha and beta), and the furanose form (alpha and beta).
How is furanose formed?
Furanose form can exist in an equilibrium distribution between a cyclic hemiacetal or cyclic hemiketal and an uncyclized free aldehyde or ketone, respectively. The anomeric carbon produced during cyclic hemiacetal formation is bonded to two oxygens.