Is aluminum a negative electrode?

Michael Henderson
Published Mar 03, 2026
Is aluminum a negative electrode?
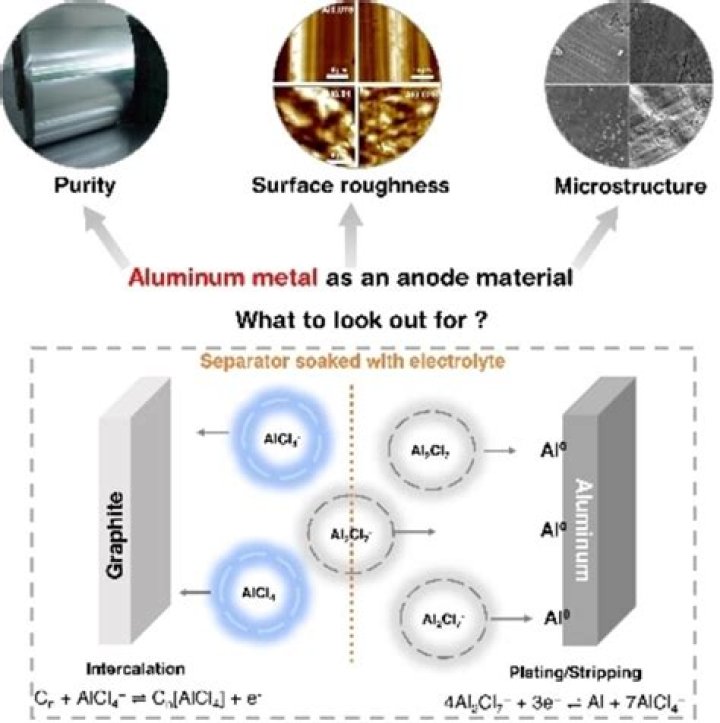
In search of new non-carbonaceous anode materials for lithium ion batteries, aluminum has been tested as a possible candidate. Despite a huge loss in capacity due to volume changes in the electrode upon cycling, aluminum appears as a good material as a negative electrode for lithium ion batteries.
What is the charge on a aluminium ion?
+3
Aluminum cation
| PubChem CID | 104727 |
|---|---|
| Molecular Weight | 26.981538 |
| Dates | Modify 2021-12-05 Create 2004-09-16 |
| Description | Aluminium(3+) is an aluminium cation that has a charge of +3. It is an aluminium cation, a monoatomic trication and a monoatomic aluminium. ChEBI |
What is aluminum-ion?
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions provide energy by flowing from the positive electrode of the battery, the anode, to the negative electrode, the cathode. When recharging, aluminium ions return to the negative electrode, and can exchange three electrons per ion.
Why is aluminium used in batteries?
The benefit of these materials over lithium is their stability. This makes for a safer battery, with faster charging time and denser energy storage capacity. Aluminum-ion batteries charge faster than their lithium-ion counterparts. Lithium-ions can only move one at a time through the battery in recharging.
Why do aluminium ions collect at the negative electrode?
Extracting Aluminium Using Electrolysis Aluminium oxide has a high melting point and so is mixed with cryolite to lower it. The aluminium oxide mixture is then melted so that the ions are free to move. The Al3+ ions are attracted to the cathode while the O2- ions are attracted to the anode.
Is aluminum-ion a metal?
On the other hand, aluminum is the most abundant metal in the earth’s crust. The discovery of inorganic materials with high aluminum-ion mobility—usable as solid electrolytes or intercalation electrodes—is an innovative and required leap forward in the field of rechargeable high-valent ion batteries.
Is aluminum a negative ion?
Aluminum, a member of the IIIA family, loses three electrons to form a 3+ cation. The halogens (VIIA elements) all have seven valence electrons. All the halogens gain a single electron to fill their valence energy level….Positive and Negative Ions: Cations and Anions.
| Family | Element | Ion Name |
|---|---|---|
| IIIA | Aluminum | Aluminum cation |
Is aluminum ion a cation or anion?
List of Ions in the CCCBDB
| Species | Name | charge |
|---|---|---|
| Al+ | Aluminum atom cation | 1 |
| Sc+ | Scandium cation | 1 |
| Ga- | Gallium atom anion | -1 |
| Ga+ | Gallium atom cation | 1 |
Is aluminum-ion a cation or anion?
What type of ion will aluminum form?
+3 ion
Aluminum is in the fifth column and therefore has 5 electrons in its outermost shell. It would tend to lose three electrons and form a +3 ion.
Is aluminium a cation or anion?
List of Ions in the CCCBDB
| Species | Name | charge |
|---|---|---|
| Al- | Aluminum atom anion | -1 |
| Al+ | Aluminum atom cation | 1 |
| Sc+ | Scandium cation | 1 |
| Ga- | Gallium atom anion | -1 |
Are the Aluminium ions oxidised or reduced when they reach the negative electrode?
Positively charged ions move to the negative electrode during electrolysis. They receive electrons and are reduced . Negatively charged ions move to the positive electrode during electrolysis. They lose electrons and are oxidised .