How is calcium oxide used in flue gas desulfurization?

Sarah Cherry
Published Mar 01, 2026
How is calcium oxide used in flue gas desulfurization?
Calcium oxide (CaO – quicklime) or calcium hydroxide (Ca(OH)2 – hydrated lime), and calcium carbonate (CaCO3 – limestone or chalk) can each be used to neutralise the acidic gases and remove sulfur dioxide from flue gases.
What is the process of flue gas desulphurisation?
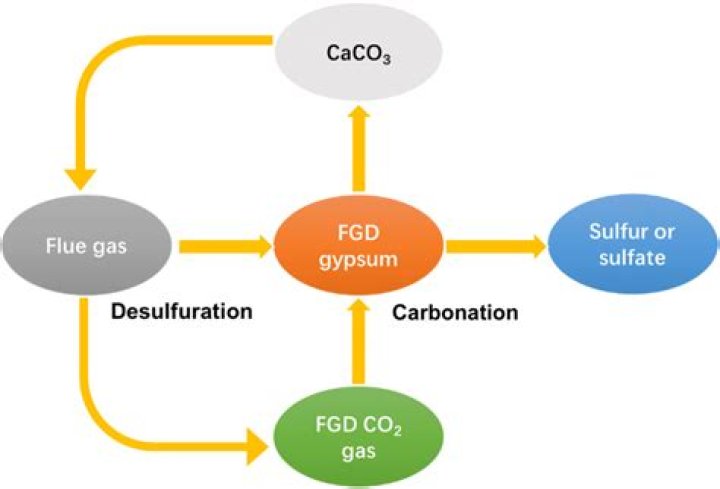
In a flue gas desulphurisation system (FGD), sulphur compounds are removed from the exhaust emissions of fossil-fuelled power stations. This is done by means of an industrial process through the addition of absorbents. In this method, the flue gases are steam-saturated with the absorbent in aqueous solution.
How much does flue gas desulfurization cost?
Total capital costs vary from $60 to $100/kw and $94 to $145/kw, respectively, for the limestone and sodium solution scrubbing systems for this model plant. Higher capital costs are often required for application of FGD systems to existing plants than for application to sim- ilar new plants.
How is SO2 removed from power plant flue gas?
The FDG or SO2 scrubbing process typically uses a calcium or sodium based alkaline reagent. The reagent is injected in the flue gas in a spray tower or directly into the duct. The SO2 is absorbed, neutralized and/or oxidized by the alkaline reagent into a solid compound, either calcium or sodium sulfate.
Why is Limewater used in wet scrubbers?
Wet scrubbing primarily uses magnesium-enhanced lime (containing 3-8% magnesium oxide) because it provides high alkalinity to increase SO2 removal capacity and reduce scaling potential.
What is meant by flue gas desulphurisation FGD?
Flue-gas desulfurization (FGD) is the removal process of sulfur dioxide (SO2) from flue gas emissions, often chemically. Sulfur dioxide in gases is produced by the combustion of fossil fuels and many industrial processes such as gasoline refining as well as cement, paper, glass, steel, iron and copper production.
What is the importance of desulfurization?
These processes are of great industrial and environmental importance as they provide the bulk of sulfur used in industry (Claus process and Contact process), sulfur-free compounds that could otherwise not be used in a great number of catalytic processes, and also reduce the release of harmful sulfur compounds into the …
How do you remove sulfur dioxide?
It is possible to scrub sulfur dioxide by using a cold solution of sodium sulfite; this forms a sodium hydrogen sulfite solution. By heating this solution it is possible to reverse the reaction to form sulfur dioxide and the sodium sulfite solution.
Do scrubbers remove SO2?
How does a scrubber work? Once sulfur is burned and produces SO2, the exhaust gas passes through the scrubber where a spray mixture of limestone (or other chemical reagent) and water reacts with the SO2. The reaction enables the SO2 to be removed before it’s released into the atmosphere.
Can scrubber remove CO2?
Carbon dioxide scrubbers are also used in controlled atmosphere They have also been researched for carbon capture and storage as a means of combating global warming. A CO2 Scrubber removes carbon dioxide from the air.
Where is flue gas desulfurization used?
Flue Gas Desulfurization Systems: Scrubbers They are used in coal-and-oil fired combustion units including utility and industrial boilers, municipal and medical waste incinerators, petroleum refineries, cement and lime kilns, metal smelters, and sulphuric acid plants.
What is the wet flue gas desulfurization process?
Several wet flue gas desulfurization (FGD) processes are based on the absorption of sulfur dioxide in caustic suspension or absorption liquor. The dissolved sulfur dioxide, which we will refer to as S (IV), is neutralized by the additive, commonly lime or limestone [1].
Where does sulfur dioxide come from when it is desulfurized?
Before flue gas desulfurization was installed, the emissions from the Four Corners Generating Station in New Mexico contained a significant amount of sulfur dioxide. 2) from exhaust flue gases of fossil-fuel power plants, and from the emissions of other sulfur oxide emitting processes such as waste incineration .
Can “flue gas” desulfurization be used on board ships?
Different methods for “flue gas” desulfurization are used on conventional coal and oil-fired power plants, but, due to cost and space requirements, the methods have not been used on board ships.
What happens when oxygen is added to the flue gas?
However, oxygen in the flue gas has a major impact on chemistry and, in particular, on by-product formation. Aqueous bisulfite and sulfite ions react with oxygen to produce sulfate ions (SO 4 -2 ).