How does salt affect osmosis?

Christopher Anderson
Published Mar 23, 2026
How does salt affect osmosis?
Salt triggers osmosis by attracting the water and causing it to move toward it, across the membrane. Salt is a solute. When you add water to a solute, it diffuses, spreading out the concentration of salt, creating a solution.
What happens to human cell in salt water?
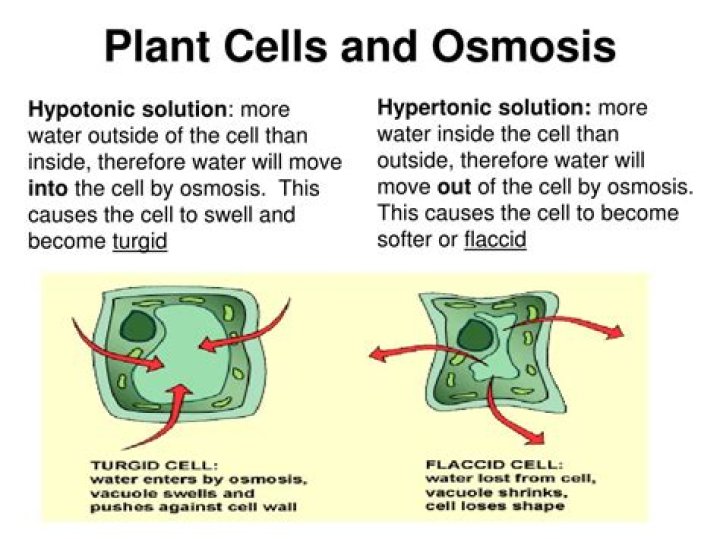
If the solution is hypotonic— having a lower concentration of solute and higher concentration of water molecules—the cells will gain water and swell. Saltwater from the ocean is hypertonic to the cells of the human body since it has more salt in it. Cells, as a result, lose water and shrink (see Figure L3. 2).
What happens to cells if they are put in a salty environment?
When plant cells are put in really salty water, water diffuses/moves out of the cell and the central vacuole shrinks. When animal cells are put in salty water, water diffuses/moves out of the cell and the cell shrivels up.
How can sugar or salt affect osmosis?
The water concentration can be thought of as the proportion of a solution that is water. Solutions with a high concentration of solute molecules, such as sugars or salts, have a low concentration of water molecules and vice versa.
How do we use osmosis in everyday life?
To better explain this phenomenon, we have listed a few very good examples of osmosis that we encounter in everyday life.
- Fish Absorb Water Through Their Skin and Gills.
- Red Blood Cells Placed Into Freshwater.
- Salt on Slugs.
- Plants Absorb Water From The Soil.
- Potato In Sugar Solution.
- Raisin In Water.
Why can we not drink salt water?
Why can’t people drink sea water? Seawater is toxic to humans because your body is unable to get rid of the salt that comes from seawater. Your body’s kidneys normally remove excess salt by producing urine, but the body needs freshwater to dilute the salt in your body for the kidneys to work properly.
Are humans hypertonic to salt water?
When we take in too much salt, our kidneys must get rid of the excess salt through our urine. Human blood has a salinity of 9, which means there are 9 grams of salt and 991 grams of water in every 1,000 grams of fluid. Saltwater is considered a hypertonic fluid, which means it contains more salt than human blood.
What would happen if a red blood cell was placed in salt water?
Red blood cells placed in a solution with a lower water concentration compared to their contents (eg 1.7 per cent salt solution) will lose water by osmosis and shrink. Water will diffuse from a higher water concentration inside the cell to a lower water concentration outside the cell.
Why dont plant cells burst if a lot of water diffuses into them?
Plant cells have a strong cellulose cell wall outside the cell membrane. The cell wall is fully permeable to all molecules and supports the cell and stops it bursting when it gains water by osmosis.
Why did plant in salt water wither?
When salt concentrations in the soil are high, the movement of water from the soil to the root is slowed down. When the salt concentrations in the soil are higher than inside the root cells, the soil will draw water from the root, and the plant will wilt and die.
What is Exoosmosis?
Exosmosis meaning The passage of a fluid through a semipermeable membrane toward a solution of lower concentration, especially the passage of water through a cell membrane into the surrounding medium.
Does sugar speed up osmosis?
As the concentration of sugar solution increases, change in mass of the potato decreases. That is the rate of osmosis decreases with the decrease in concentration of water molecules. The concentration inside and outside of the potato will be the same so no osmosis will be occurring.
What happens when there is too much salt in a cell?
Water in cells moves toward the highest concentration of salt. If there is more salt in a cell than outside it, the water will move through the membrane into the cell, causing it to increase in size, swelling up as the water fills the cell in its imperative to combine with the salt.
What happens to your body when you use osmosis?
If you’re consuming seawater, the results of osmosis are spectacularly disastrous. Remember the salinity of seawater is almost four times that of our bodily fluids. If gone unchecked, the net transfer of water from the inside of your cells to the outside will cause the cells to shrink considerably –…
What happens when you add salt to water?
Salt is a solute. When you add water to a solute, it diffuses, spreading out the concentration of salt, creating a solution. If the concentration of salt inside a cell is the same as the concentration of salt outside the cell, the water level will stay the same,…
What happens to plant cells in salt water?
If a higher concentration of salt is placed outside of the cell membrane, the water will leave the cell to bond with it. The loss of water from this movement causes plant cells to shrink and wilt. This is why salt can kill plants; it leaches the water from the cells.
Water in cells moves toward the highest concentration of salt. If there is more salt in a cell than outside it, the water will move through the membrane into the cell, causing it to increase in size, swelling up as the water fills the cell in its imperative to combine with the salt.
If you’re consuming seawater, the results of osmosis are spectacularly disastrous. Remember the salinity of seawater is almost four times that of our bodily fluids. If gone unchecked, the net transfer of water from the inside of your cells to the outside will cause the cells to shrink considerably –…
Salt is a solute. When you add water to a solute, it diffuses, spreading out the concentration of salt, creating a solution. If the concentration of salt inside a cell is the same as the concentration of salt outside the cell, the water level will stay the same,…
If a higher concentration of salt is placed outside of the cell membrane, the water will leave the cell to bond with it. The loss of water from this movement causes plant cells to shrink and wilt. This is why salt can kill plants; it leaches the water from the cells.