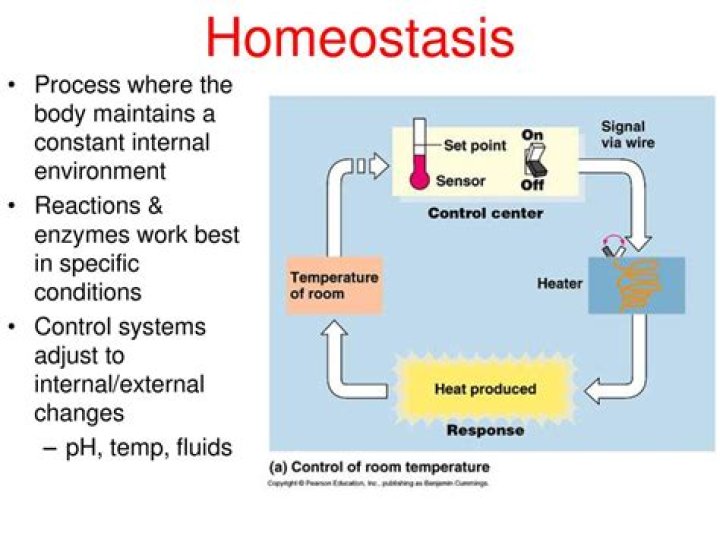
How does blood pH maintain homeostasis

Michael Henderson
Published Apr 08, 2026
The most important way that the pH of the blood is kept relatively constant is by buffers dissolved in the blood. Other organs help enhance the homeostatic function of the buffers. The kidneys help remove excess chemicals from the blood, as discussed in the Kidney Dialysis tutorial.
How the body maintains homeostasis of blood pH?
Body fluid pH is strictly maintained by buffering systems, efflux across plasma membrane, and acid excretion. Monocarboxylate transporter (MCT) and Na+/H+ exchanger (NHE) contribute to proton extrusion from the cytosol to the extracellular space.
What are the three major mechanisms of pH regulation?
There are three important mechanisms the body uses to regulate pH. The first is a chemical buffer, the second line of defense is the respiratory system, and last, is the urinary system. These three mechanisms work together to keep body pH within that narrow range.
Why is pH important in maintaining homeostasis?
The pH value is important in maintaining homeostasis because it provides the correct balance of hydrogen ions in solution.Why is it important to maintain the pH of blood and tissue fluid?
Why is it important to maintain the pH of blood and tissue fluids within normal limits? The structure and function of macromolecules are pH dependent and Slight deviations from normal pH can shut down metabolic pathways.
How does pH affect cells and cellular processes?
Changes in intracellular pH can potentially affect virtually all cellular processes, including metabolism, membrane potential, cell growth, movement of substances across the surface membrane, state of polymerization of the cytoskeleton and ability to contract in muscle cells.
How do cells maintain pH?
Cells adjust their pHi accordingly upon an increase in acidity or basicity, usually with the help of CO2 or HCO3– sensors present in the membrane of the cell. These sensors can permit H+ to pass through the cell membrane accordingly, allowing for pHi to be interrelated with extracellular pH in this respect.
Why is pH within the cell so important to cellular function?
pH is important because the enzymes that catalyze the chemical reactions of life require a specific pH in order to function. … This ensures that the cells’ environments are maintained at the proper pH, in order for their biomolecules to maintain their functionality, and so that their enzymes can function properly.How is the pH in the bioreactor controlled?
In the beginning of the culture, pH is regulated in the range 7.0 – 7.4 by playing on the bicarbonate equilibrium. CO2 is added in the sparger to increase dissolved CO2 and decrease pH. Alternatively, air is added in the sparger to strip the dissolved CO2 out and increase pH.
What causes changes in pH of blood?Causes of blood pH changes. Metabolic changes in blood pH can occur as a result of kidney conditions or problems. Respiratory changes relate to how the lungs are working. When a change happens in one direction, there are mechanisms to move the acid-base balance the other way.
Article first time published onHow does the body respond to a change in pH?
A slight change in pH in the spinal fluid and cerebral fluid during acidosis causes a reduction in affinity of hemoglobin for oxygen, reducing the critical oxygen supply to brain cells. … During alkalosis, or a rise in pH, blood vessels constrict and thereby reduce the supply of blood and oxygen to brain cells.
How does pH affect cell membrane?
Membrane lipids are directly affected by pH, due to their acido-basic properties. pH change can induce lipid vesicle migration and global deformation. pH change can cause polarization in phase-separated membrane of GUVs. Localized pH heterogeneities can induce local dynamical membrane deformations.
Why is pH control important in cell culture operations?
pH control is essential to optimizing biopharma’s key quality parameters, and ensuring final product quality and yield. … Metabolic changes within micro-organisms can change the pH of their environment, as can changes in process conditions.
How is pH regulated in cell cultures?
The most common buffer used in mammalian cell culture is sodium bicarbonate. The sodium bicarbonate buffered media becomes sensitive to carbonic acid formed by the concentration of CO2. As long as the level of CO2 is controlled, the pH of the media can be maintained.
What is the importance of pH adjustment in culture media?
Different types of culture media are designed for different types of microorganisms and applications. pH affects the physical appearance of culture media and their ability to grow microorganisms. Most bacteria grow in pH 6.5 – 7.0 while animal cells thrive in pH 7.2 – 7.4.
How does pH affect biological activities and functions?
pH is biologically important because it affects the structure and activity of macromolecules. pH is important in homeostatic processes. For example, most animals breathe not because they lack oxygen, but because CO2 buildup in the blood increases the blood acidity beyond normal levels.
What happens if blood becomes acidic?
As blood pH drops (becomes more acidic), the parts of the brain that regulate breathing are stimulated to produce faster and deeper breathing (respiratory compensation). Breathing faster and deeper increases the amount of carbon dioxide exhaled, which raises the blood pH back toward normal.
What actions affect blood pH?
The lower the pH, the more acidic the blood. A variety of factors affect blood pH including what is ingested, vomiting, diarrhea, lung function, endocrine function, kidney function, and urinary tract infection.
What factors maintain the normal blood pH?
This analysis has revealed three independent variables that regulate pH in blood plasma. These variables are carbon dioxide, relative electrolyte concentrations, and total weak acid concentrations. All changes in blood pH, in health and in disease, occur through changes in these three variables.
How do you maintain the pH level in your body?
To balance your pH and optimise your health and wellbeing we recommend eating plenty of the foods from the alkaline list and limiting the intake of acidic foods. Another way to stabilise pH is to drink alkaline water. Normal drinking water sits at a pH of 7 – neutral.
Why is pH so important in the human body?
It is important for us to assist our body in creating and maintaining a healthy pH ratio of 70:30, alkaline to acid. Most diseases, illnesses, and bad bacteria thrive in an over acidic environment. When pH levels are unbalanced, it is mostly in the case of being too acidic.
Does pH affect diffusion?
Diffusion coefficients were determined by means of numerical solutions of Fick’s laws with appropriate boundary conditions. It has been found that the pH of the medium strongly influences diffusion.
Why does pH affect cell membrane permeability?
As the pH got higher the absorbance of light got lower. We concluded that high pH makes cell membranes become less permeable, allowing less molecules to pass through.
How does pH affect the phospholipid bilayer?
The change in the pH of the solution induces changes in electrical charge of the membrane due to the variations in acid–base equilibrium of the groups present in the lipid molecule. At a certain pH value, the number of positive and negative groups is equal.
How do buffers regulate pH in cells?
Buffers are chemicals that can easily release or take up hydrogen ions in a liquid, meaning they are able resist a change in pH by controlling how many free-floating hydrogen ions there are. The pH scale ranges from 0 to 14. A pH from 0 to 7 is considered acidic and a pH from 7 to 14 is considered basic.
Why is pH important in bioreactor?
Controlling cell culture pH in bioreactors is critical to successful manufacturing. Most culture needs to be maintained in a specific narrow range of pH for maximum productivity. … Several factors affect pH including temperature, cell growth, lactic acid, and CO2 levels.
What is the pH value of human blood *?
Blood is normally slightly basic, with a normal pH range of about 7.35 to 7.45. Usually the body maintains the pH of blood close to 7.40.
What molecules help maintain a stable internal pH?
The buffer that maintains the pH of human blood involves carbonic acid (H2CO3), bicarbonate ion (HCO3–), and carbon dioxide (CO2).



