How does adenylate kinase make ATP?

Daniel Johnson
Published Mar 21, 2026
How does adenylate kinase make ATP?
Adenylate kinase (AK) is a ubiquitous and abundant enzyme catalyzing the phosphoryl transfer between two adenosine diphosphate (ADP) molecules to yield adenosine triphosphate (ATP) and adenosine monophosphate (AMP). AK can be used to regenerate ATP from the AMP produced in the luciferase reaction.
What pathway is catalysed by adenylate kinase?
Recent studies provide new evidence of the unique energetic, metabolic monitoring and signaling role played by the ubiquitous enzyme adenylate kinase which catalyzes the nucleotide phosphoryl exchange reaction 2ADP ↔ ATP + AMP, critical in cell life [1–11].
What is the mechanism of protein kinases?
Protein kinases (PTKs) are enzymes that regulate the biological activity of proteins by phosphorylation of specific amino acids with ATP as the source of phosphate, thereby inducing a conformational change from an inactive to an active form of the protein.
How are kinases activated?
Activation is mediated by binding of cyclic AMP to the regulatory subunits, which causes the release of the catalytic subunits. cAPK is primarily a cytoplasmic protein, but upon activation it can migrate to the nucleus, where it phosphorylates proteins important for gene regulation. Domain movements in protein kinases.
What is the role of adenylate kinase?
Adenylate kinase (Adk) is an essential housekeeping enzyme (9) that controls the energy balance in cells by catalyzing the reversible interconversion of ATP and AMP into two ADP molecules.
What is the purpose of adenylate kinase?
Adenylate kinase (AK) is the critical enzyme in the metabolic monitoring of cellular adenine nucleotide homeostasis. It also directs AK→ AMP→ AMPK signaling controlling cell cycle and proliferation, and ATP energy transfer from mitochondria to distribute energy among cellular processes.
What happens when protein kinase is activated?
Protein kinase A (PKA) is activated by the binding of cyclic AMP (cAMP), which causes it to undergo a conformational change. As previously mentioned, PKA then goes on to phosphoylate other proteins in a phosphorylation cascade (which required ATP hydrolysis).
How do kinase inhibitors work?
Tyrosine kinase inhibitors (TKIs) block chemical messengers (enzymes) called tyrosine kinases. Tyrosine kinases help to send growth signals in cells, so blocking them stops the cell growing and dividing. Cancer growth blockers can block one type of tyrosine kinase or more than one type.
How does phosphorylation by kinases control protein activity?
What activates protein kinase?
Protein kinase A (PKA) is activated by the binding of cyclic AMP (cAMP), which causes it to undergo a conformational change. The alpha subunit then binds to adenylyl cyclase, which converts ATP into cAMP. cAMP then binds to protein kinase A, which activates it.
What does adenylate kinase do in glycolysis?
Adenylate kinase (EC 2.7. 4.3) (also known as ADK or myokinase) is a phosphotransferase enzyme that catalyzes the interconversion of the various adenosine phosphates (ATP, ADP, and AMP). By constantly monitoring phosphate nucleotide levels inside the cell, ADK plays an important role in cellular energy homeostasis.
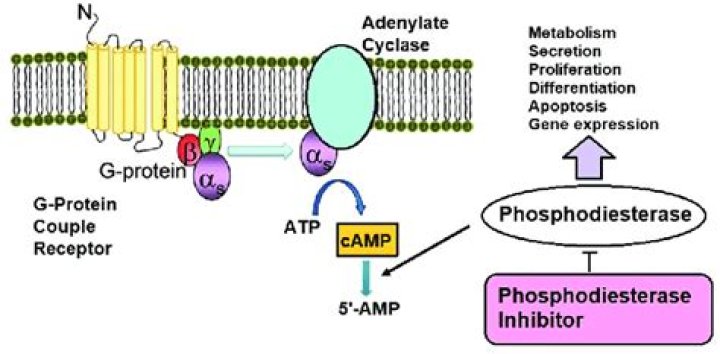
What does adenylate cyclase do?
Adenylyl cyclase is the sole enzyme to synthesize cyclic AMP (cAMP), a key second messenger that regulates diverse physiological responses including sugar and lipid metabolism, olfaction, and cell growth and differentiation.
What is the function of adenylate kinase (ADK)?
Adenylate kinase ( EC 2.7.4.3) (also known as ADK or myokinase) is a phosphotransferase enzyme that catalyzes the interconversion of adenine nucleotides (ATP, ADP, and AMP). By constantly monitoring phosphate nucleotide levels inside the cell, ADK plays an important role in cellular energy homeostasis.
What is adenylate kinase deficiency in erythrocyte?
Adenylate kinase deficiency in the erythrocyte is associated with hemolytic anemia. This is a rare hereditary erythroenzymopathy that, in some cases, is associated with mental retardation and psychomotor impairment. At least two patients have exhibited neonatal icterus and splenomegaly and required blood transfusions due to this deficiency.
How is sub-cellular localization of ADK enzymes done?
Sub-cellular localization of the ADK enzymes is done by including a targeting sequence in the protein. Each isoform also has different preference for NTP’s. Some will only use ATP, whereas others will accept GTP, UTP, and CTP as the phosphoryl carrier. Some of these isoforms prefer other NTP’s entirely.
What factors affect adenine nucleotide activity?
Common factors that influence adenine nucleotide levels, and therefore ADK activity are exercise, stress, changes in hormone levels, and diet. It facilitates decoding of cellular information by catalyzing nucleotide exchange in the intimate “sensing zone” of metabolic sensors.



