How does acid rain affect water cycle?

Michael Henderson
Published Feb 09, 2026
How does acid rain affect water cycle?
As it flows through the soil, acidic rain water can leach aluminum from soil clay particles and then flow into streams and lakes. The more acid that is introduced to the ecosystem, the more aluminum is released. Some types of plants and animals are able to tolerate acidic waters and moderate amounts of aluminum.
What cycles involve acid rain?
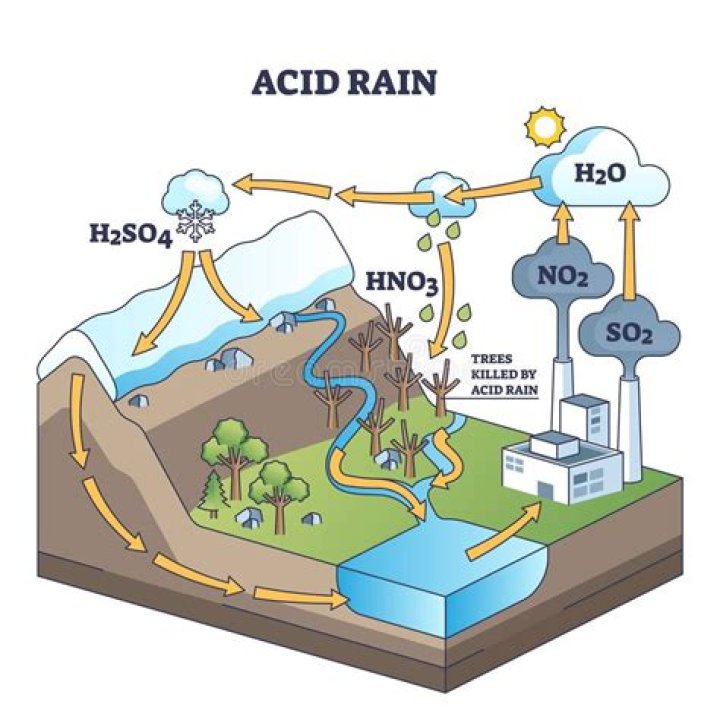
1. H2SO4 dissociates in water to give 2 H+ and SO42- . Thus, the cycles of nitrogen and sulfur interact to form acid rain.
How does the water cycle and the carbon cycle work to make acid rain?
The movement of carbon from the atmosphere to the lithosphere (rocks) begins with rain. Atmospheric carbon combines with water to form a weak acid—carbonic acid—that falls to the surface in rain. The acid dissolves rocks—a process called chemical weathering—and releases calcium, magnesium, potassium, or sodium ions.
How does acid rain affect the nitrogen cycle?
The release of nitric oxides into the air in large quantities causes smog and acid rain that pollutes the atmosphere, soil and water and affects plants and animals. Also, soils become significantly more acidic, as do stream systems and lakes as the nitrogen feeds into the water supply.
How does acid rain get into water?
When acid rain reaches Earth, it flows across the surface in runoff water, enters water systems, and sinks into the soil. Caused when rain droplets absorb air pollution like sulfur and nitrogen oxides, acid rain weakens trees by dissolving nutrients in the soil before plants can use them.
What is acid rain Slideshare?
Acid Rain is the Precipitation that has a pH of less than that of natural rainwater (which is about 5.6 due to dissolved carbon dioxide) It is formed when sulphur dioxides and nitrogen oxides, as gases or fine particles in the atmosphere, combine with water vapour and precipitate as sulphuric acid or nitric acid in …
What is acid rain and how is it formed discuss the major impacts of acid rain and the measures to control it?
Alternative energy sources should be used, such as solar and wind power. Renewable sources of energy are helping to reduce acid rain, as they produce much fewer emissions….What are three ways to reduce acid rain?
| CHEMISTRY Related Links | |
|---|---|
| Ore Definition | Atomic Mass Of Potassium |
| Aniline Structure | Atomic Mass Of Zinc |
Can you drink acid rain?
Humans are affected when we breathe in air pollution, this can cause breathing problems, and even cancer. Drinking water which has been contaminated with acid rain can cause brain damage over time.
What is the main source of acid rain?
Power plants release the majority of sulfur dioxide and much of the nitrogen oxides when they burn fossil fuels, such as coal, to produce electricity. In addition, the exhaust from cars, trucks, and buses releases nitrogen oxides and sulfur dioxide into the air. These pollutants cause acid rain.
What are the advantages and disadvantages of acid rain?
Acid rain affects the fresh water ponds and lakes and destroys the aquatic life as some species of fishes are rare and may be extinct. it can affect the trees particularly those that are high altitude. it can damage historical monuments and buildings. The buildings can be found with sulfuric acid.